
US-based biotechnology firm Celgene has reported positive top-line results from the Phase III clinical trial (SUNBEAM) of oral ozanimod for the treatment of patients with relapsing multiple sclerosis (RMS).
Ozanimod is a new, oral, selective, sphingosine 1-phosphate 1 (S1PR1) and 5 (S1PR5) receptor modulator currently being developed for immune-inflammatory diseases such as relapsing multiple sclerosis, ulcerative colitis and Crohn’s disease.
The results showed that both drug doses have demonstrated statistically significant and clinically meaningful improvements for the primary endpoint of annualised relapse rate (ARR).
The multicentre, randomised, double-blind, double-dummy, active-controlled Phase III trial evaluated the efficacy, safety and tolerability of 0.5mg and 1mg oral doses of ozanimod over a 12-month treatment period in 1,346 patients.
See Also:
The drug was assessed against weekly intramuscular interferon beta-1a (Avonex).
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataCelgene inflammation and immunology president Scott Smith said: “People living with multiple sclerosis need additional therapies and we are pleased that oral ozanimod showed meaningful improvements across primary and measured secondary endpoints in this study.
“We look forward to data from the confirmatory Phase III Radiance trial in the second quarter as we advance toward planned regulatory submissions by the year-end.”
The data was found to be encouraging for the measured secondary endpoints such as the number of gadolinium-enhancing MRI lesions and the number of new or enlarging T2 MRI lesions at month 12.
It was reported that the overall safety and tolerability profile of the drug was consistent with previously conducted Phase II RMS (RADIANCE) and Phase II ulcerative colitis (TOUCHSTONE) trials.
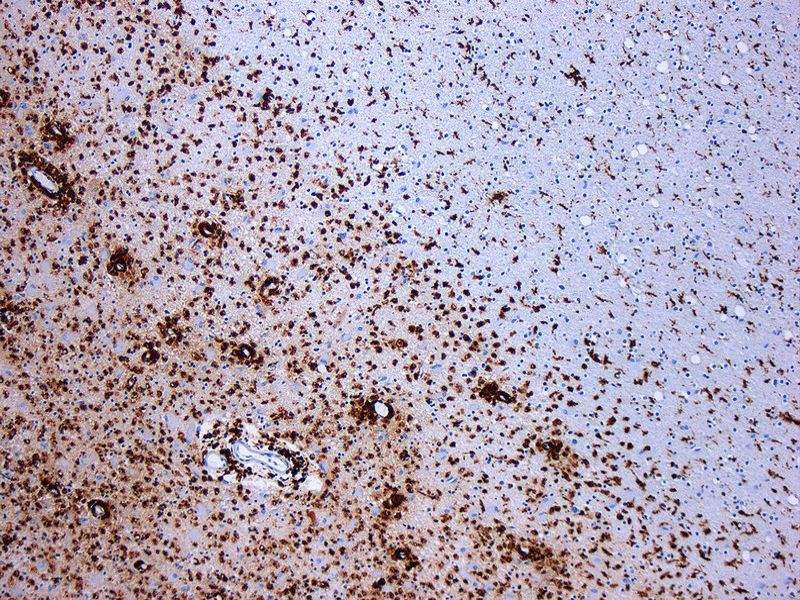
Image: Photomicrograph of a demyelinating MS-Lesion. Photo: courtesy of Marvin 101 / Wikipedia.