
A new Phase I/II clinical trial of MV-CHIKV sponsored by the National Institutes of Health’s (NIH) National Institute of Allergy and Infectious Diseases (NIAID) for the prevention of the chikungunya virus has begun enrolling healthy adult subjects at three US clinical centres.
The trial is being conducted at various NIAID-funded vaccine and treatment evaluation units.
Developed by Austrian-based biotechnology company Themis Bioscience, MV-CHIKV is an experimental candidate of the measles vaccine virus that is altered to generate chikungunya virus proteins.
Upon entering a human cell, the vaccine activates both measles and chikungunya protein production, against which the immune system develops antibodies, which may have the potential to prevent future chikungunya infection.
See Also:
NIAID director Anthony Fauci said: “Chikungunya virus can cause debilitating joint pain that can last for months or even longer.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalData“A vaccine to prevent infection with this virus would be of considerable benefit to people living in the more than 60 countries where chikungunya transmission has occurred, as well as travellers to those countries.”
The trial will recruit a total of 180 healthy volunteers aged 18 to 45 at the University of Iowa in Iowa City, Baylor College of Medicine in Houston and Emory University in Atlanta.
The subjects will be administered with two injections of either low-dose or high-dose MV-CHIKV or placebo, and will be monitored for adverse reactions over a period of eight to 13 months.
A Phase I trial conducted in 2014 by Themis Bioscience demonstrated the safety of MV-CHIKV and its ability to trigger an immune response.
The experimental vaccine is being further studied in a Phase II trial in Europe.
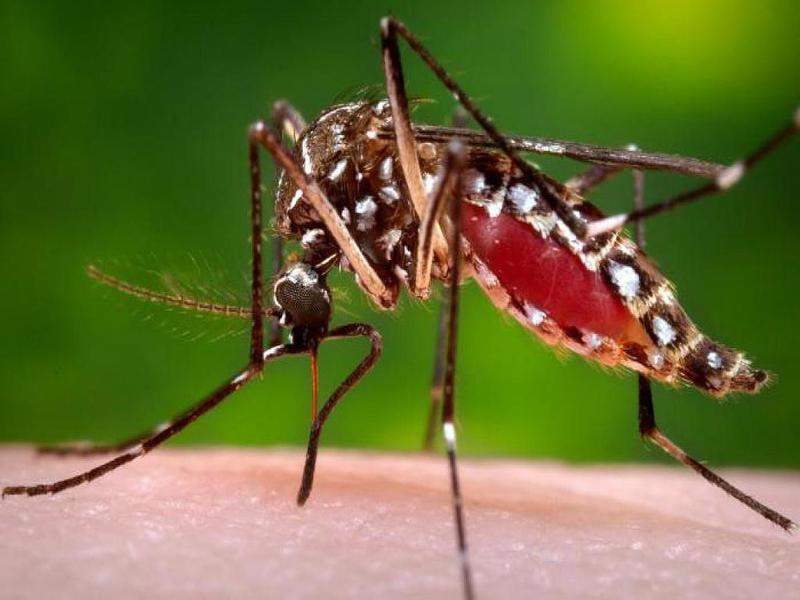
Image: A female Aedes aegypti mosquito, one of the two species that spreads chikungunya virus. Photo: courtesy of Centres for Disease Control and Prevention (CDC).