
The National University of Ireland (NUI) Galway has initiated the Phase I clinical trial of daratumumab combination therapy in Irish patients for the treatment of multiple myeloma.
The patients in Ireland are the first to receive this new combination therapy.
Daratumumab (DARA) is a therapy being developed to treat multiple myeloma and is approved for the treatment of relapsed patients.
The Phase I trial is designed to evaluate DARA in combination with a standard care chemotherapy, comprising the drugs cyclophosphamide and bortezomib (CyBorD) to treat newly diagnosed patients.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataSee Also:
BCNI director, lead investigator and NUI Galway consultant haematologist Michael Dwyer said: "This new trial, a first for BCNI, is another step forward in developing new treatment options for patients living with multiple myeloma.
"The study is the result of collaborations across a broad range of partners, including NUI Galway, Cancer Trials Ireland, the Irish Cancer Society, Science Foundation Ireland, Janssen Pharmaceuticals, the Health Research Board and BCNI investigators and staff."
The first six patients were recruited by Blood Cancer Network Ireland (BCNI) at University Hospital Galway and Cork University Hospital, with plans to extend the trial to BCNI centres in Dublin.
This trial is the first investigator-initiated trial being conducted by BCNI, as well as the first cancer clinical trial to be sponsored by NUI Galway.
A result of research efforts between BCNI scientists and Janssen pharmaceuticals, the study primarily aims to establish that the combination of cyclophosphamide treatment with DARA is more effective.
The subjects participating in the trial will receive additional benefits such as monitoring and free treatment access.
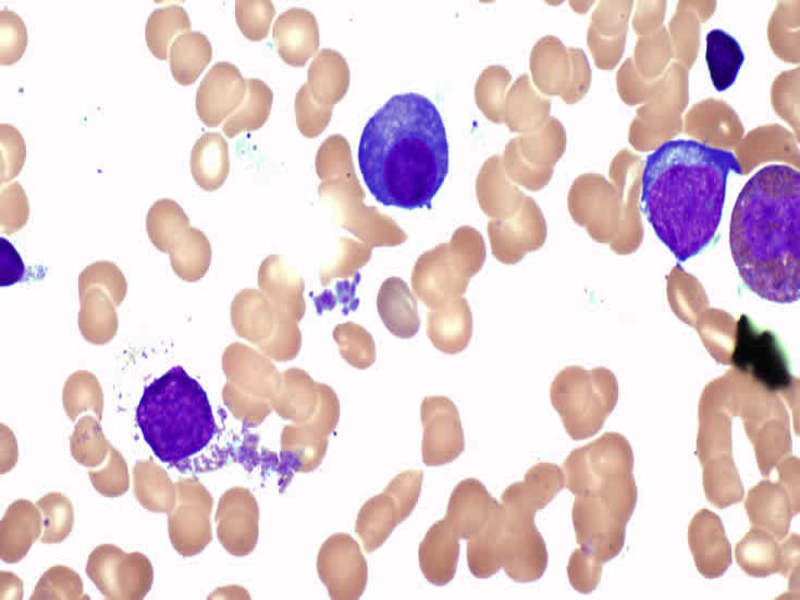
Image: Multiple myeloma cells. Photo: courtesy of NUI Galway.