Biotechnology firm Seattle Genetics has stopped the Phase III CASCADE clinical trial of vadastuximab talirine (SGN-CD33A) in older patients with frontline acute myeloid leukaemia (AML).
The move followed Independent Data Monitoring Committee’s (IDMC) review of trial data, which showed a high death rate and fatal infections in the vadastuximab talirine treatment arm.
Based on the firm’s antibody-drug conjugate (ADC) technology, vadastuximab talirine is an investigational antibody-drug conjugates (ADC) currently being developed to target CD33 protein that is found in the majority of AML and myelodysplastic syndrome (MDS) blast cells.
The randomised, double-blind, placebo-controlled CASCADE trial is designed to compare the combination of vadastuximab talirine with the hypomethylating agents (HMAs) such as azacitidine or decitabine to HMA alone in patients newly diagnosed with AML.
See Also:
Seattle Genetics president and CEO Clay Siegall said: “This is a disappointing and unexpected result for the CASCADE trial. Patient safety is our highest priority, and we will closely review the data and evaluate next steps.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalData“We are enthusiastic about the many opportunities across our broad pipeline, including Adcetris (brentuximab vedotin), enfortumab vedotin (ASG-22ME) and SGN-LIV1A.”
The firm has ended patient enrolment and treatment in all the vadastuximab talirine clinical trials such as the ongoing Phase I/II trial in patients suffering from frontline high-risk MDS.
Seattle Genetics intends to closely review the data and discuss with the US Food and Drug Administration (FDA) to establish future plans for the development programme of vadastuximab talirine.
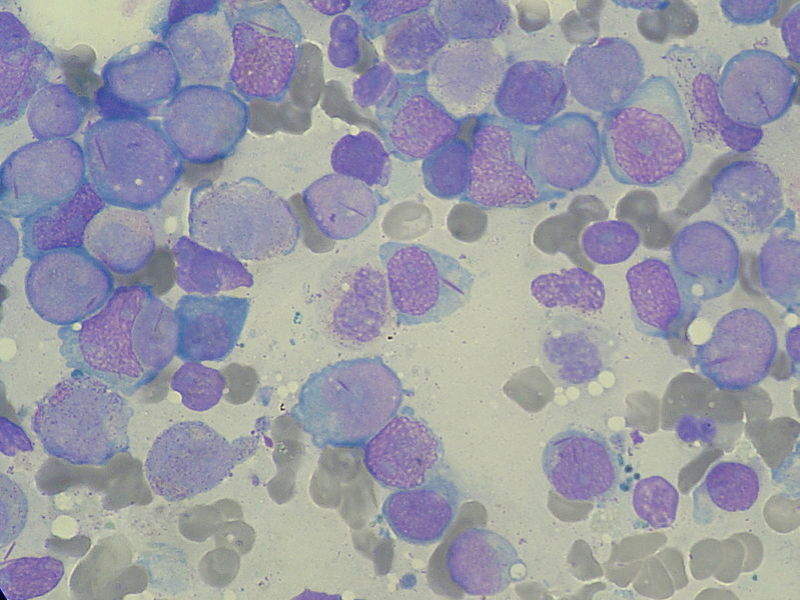
Image: Myeloblasts found in AML. Photo: courtesy of Paulo Henrique Orlandi Mourao via Wikipedia.