ICOTYDE™ (icotrokinra) is the first and only targeted oral peptide indicated for the treatment of moderate-to-severe plaque psoriasis (PsO) in adults and adolescents aged 12 years and over, weighing at least 40kg, who are suitable for systemic therapy or phototherapy.
The medicine was co‑discovered and is being advanced under a licence and collaboration agreement between Protagonist Therapeutics and Johnson & Johnson.
Under this agreement, Johnson & Johnson holds exclusive global rights to conduct phase two development and subsequent stages, and to commercialise compounds arising from the collaborative research across a broad range of indications.
The US Food and Drug Administration (FDA) approved ICOTYDE in March 2026, supported by data from the ICONIC clinical development programme.
ICOTYDE is supplied as yellowish‑orange to yellowish‑brown, oval, film‑coated tablets, each containing 200mg, for once‑daily oral administration.
Plaque psoriasis causes and symptoms
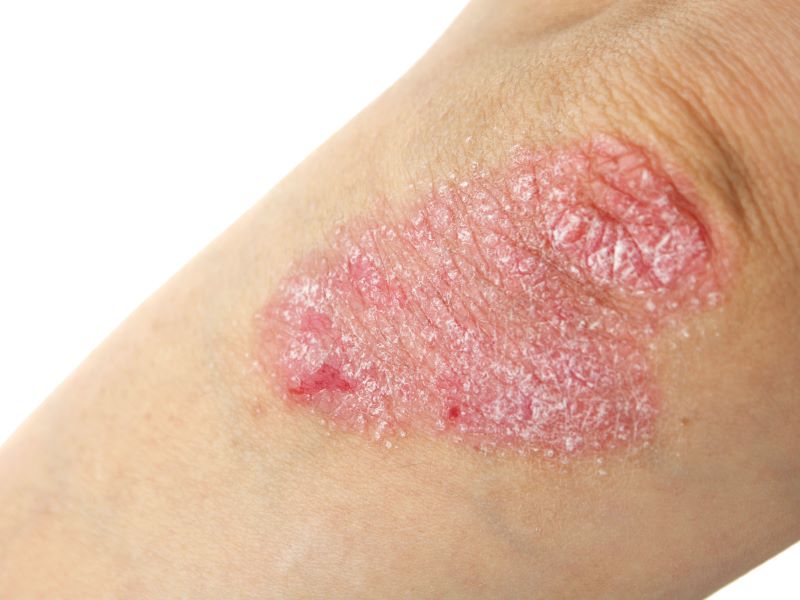
Plaque psoriasis is a chronic, immune‑mediated condition characterised by excessive skin cell production, leading to inflamed, scaly plaques that can be itchy or painful.
While its exact aetiology is not fully defined, it is broadly regarded as an immune‑mediated disease that manifests in individuals with genetic susceptibility.
The disease is characterised by lesions commonly presented as raised patches with a silvery‑white accumulation of dead skin cells. On lighter skin, plaques often appear red; on darker skin tones, they may appear purple, grey or dark brown. Although plaques can occur anywhere, they frequently involve the scalp, knees, elbows and trunk.
When psoriasis affects highly visible or sensitive areas such as the scalp, hands, feet or genitals it is associated with greater detriment to quality of life.
An estimated eight million people in the US and more than 125 million globally are affected by the disease. Around one quarter of individuals with plaque psoriasis have moderate-to-severe disease.
ICOTYDE mechanism of action
Icotrokinra is a peptide that selectively targets the interleukin‑23 receptor (IL‑23R), which is an endogenous cytokine involved in inflammatory and immune pathways.
It blocks IL‑23 from binding to its receptor and inhibits IL‑23/IL‑23R signalling in human T cells, thereby reducing the receptor‑dependent release of pro‑inflammatory cytokines.
Clinical trials on ICOTYDE
The safety and efficacy of ICOTYDEwere assessed in four Phase III, multicentre, randomised, double‑blind, placebo‑controlled clinical studies, ICONIC-ADVANCE 1, ICONIC-ADVANCE 2, ICONIC-LEAD and ICONIC-TOTAL, in patients aged 12 years and older with moderate-to-severe PsO.
Participants were randomised to either icotrokinra 200mg once daily, deucravacitinib 6mg once daily, or placebo.
The primary endpoint of the studies was Investigator’s Global Assessment (IGA) 0/1 response at week 16, defined as an IGA score of 0 (clear) or 1 (almost clear) with an improvement of at least two grades from baseline.
The co‑primary endpoint was the proportion of participants achieving at least a 90% improvement from baseline in PASI (PASI 90).
The ICONIC‑ADVANCE 1 and ICONIC‑ADVANCE 2 studies enrolled 774 and 731 adults, respectively.
In ICONIC‑ADVANCE 1, 68% of participants receiving icotrokinra achieved an IGA 0/1 response at week 16, compared with 11% on deucravacitinib or placebo.
In ICONIC‑ADVANCE 2, 70% of participants on icotrokinra achieved an IGA 0/1 response at week 16, compared with 9% on deucravacitinib or placebo.
Furthermore, icotrokinra demonstrated high rates of complete skin clearance (PASI 100) with an increase from 41% to 49% between week 24 and week 52 in ICONIC‑ADVANCE 1, and from 33% to 48% in ICONIC‑ADVANCE 2.
The ICONIC‑LEAD trial assessed the safety and efficacy of icotrokinra in 684 adults and adolescents aged 12 years and above.
At week 16, 65% of participants treated with icotrokinra achieved an IGA 0/1 response compared with 8% on placebo, and 50% achieved PASI 90 versus 4% in the placebo group.
The ICONIC‑TOTAL trial randomised 311 participants to icotrokinra or placebo. At week 16, 56.7% of those receiving icotrokinra achieved an IGA score of 0/1, compared with 5.8% in the placebo group.
Following the positive results from the ADVANCE studies, Johnson & Johnson commenced the Phase III ICONIC‑ASCEND trial (NCT06934226) in April 2025 to evaluate the efficacy and safety of icotrokinra versus placebo and Ustekinumab in participants with moderate-to-severe PsO. Additional studies are ongoing in active psoriatic arthritis, moderately to severely active ulcerative colitis, and moderately to severely active Crohn’s disease.