Lifyorli™ (relacorilant) is a selective glucocorticoid receptor antagonist (SGRA), which, along with nab-paclitaxel, is indicated for the treatment of patients with platinum‑resistant ovarian cancer (PROC).
The drug was developed by Corcept Therapeutics, a US‑based pharmaceutical company.
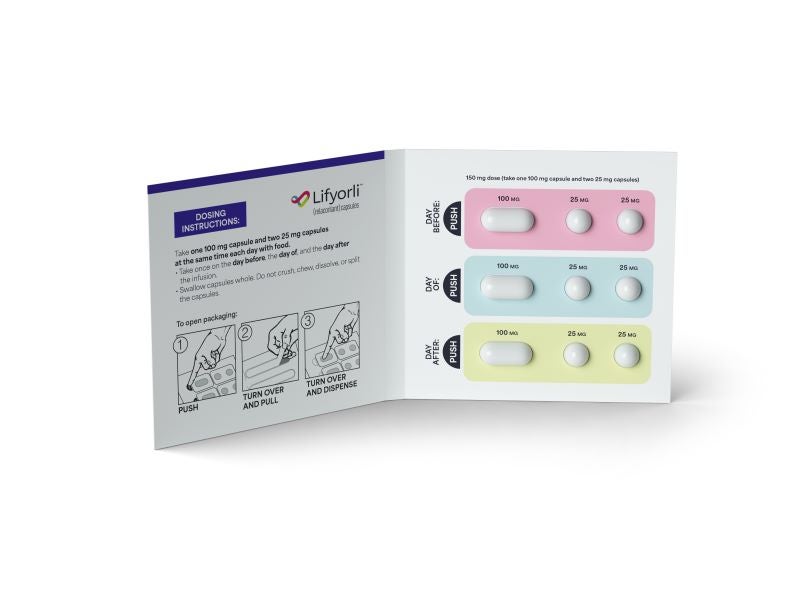
Lifyorli is available in two dosage forms and strengths, including 25mg opaque, dark brown, oval-shaped soft gelatine capsules and 100mg opaque, yellow, oblong soft gelatine capsules.
The recommended relacorilant regimen is 150mg taken orally once daily on the day before, the day of, and the day after each nab‑paclitaxel infusion.
Regulatory approvals for Lifyorli
In July 2025, Corcept Therapeutics submitted a new drug application (NDA) to the US Food and Drug Administration (FDA) for relacorilant for the treatment of patients with PROC. The FDA accepted the NDA for review in September of the same year.
In March 2026, the FDA approved Lifyorli (relacorilant) in combination with nab-paclitaxel for use in adults with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who have received one to three previous systemic treatment regimens, including at least one regimen containing bevacizumab.
Corcept Therapeutics also submitted a marketing authorisation application to the European Medicines Agency in October 2025 for relacorilant for the treatment of patients with PROC.
The drug has an orphan drug designation from both the FDA and the European Commission (EC) for the treatment of hypercortisolism and from the EC for the treatment of ovarian cancer.
Ovarian cancer causes and symptoms
Ovarian cancer is the fifth leading cause of cancer-related mortality among women, and when it recurs within six months of treatment with a platinum-based regimen is described as “platinum-resistant”.
The disease is characterised by abnormal cell proliferation in the ovaries and often presents with subtle or non-specific symptoms in the early stages, which contribute to difficulties in timely diagnosis. It is one of the most lethal gynaecological cancers and is frequently identified at an advanced stage, which complicates management and is associated with reduced overall survival.
A range of factors including genetic susceptibility, reproductive history and hormonal influences are associated with the development of ovarian cancer, highlighting the relevance of comprehensive risk assessment and appropriate screening approaches.
Therapeutic options for women with platinum-resistant disease remain limited. Each year, an estimated 20,000 women in the US, and at least a comparable number in Europe, are considered eligible to initiate a new line of treatment for PROC.
Mechanism of action
Relacorilant is a selective glucocorticoid receptor (GR) antagonist that modulates cortisol activity by binding to the GR without interacting with other endogenous hormone receptors.
In functional in vitro assays involving the mineralocorticoid receptor, relacorilant demonstrated neither agonist nor antagonist effects. In xenograft models derived from human cell lines, co-administration of relacorilant with paclitaxel increased apoptosis and enhanced anti-tumour activity.
Cortisol binding to the GR exerts immunosuppressive effects including reduced secretion of pro-inflammatory cytokines. By antagonising the GR, relacorilant may indirectly influence immune function.
Furthermore, in stimulated peripheral blood mononuclear cells, relacorilant prevented cortisol-induced reductions in tumour necrosis factor alpha and interferon gamma.
Clinical trials on Lifyorli
The US FDA approval of Lifyorli was supported by results from the pivotal ROSELLA Phase III, multicentre, open-label, active-controlled, randomised, two-arm clinical trial.
The trial investigated the safety and efficacy of relacorilant in combination with nab-paclitaxel in 381 patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who had received one to three prior systemic anti-cancer regimens, including at least one containing bevacizumab. Patients requiring chronic or frequent glucocorticoid use were excluded.
The co-primary endpoints of the study were progression-free survival and overall survival (OS).
Participants were randomised in a 1:1 ratio to receive either relacorilant plus nab-paclitaxel or nab-paclitaxel alone, without biomarker-based selection. Randomisation was stratified by number of prior treatment lines (one versus more than one) and geographic region.
In the combination arm (n=188), patients received relacorilant 150mg orally on the day before, the day of, and the day after nab-paclitaxel 80mg/m² administered intravenously on days 1, 8 and 15 of each 28-day cycle.
In the control arm (n=193), nab-paclitaxel 100mg/m² was given intravenously on days 1, 8 and 15 of each 28-day cycle.
Treatment continued until disease progression or unacceptable toxicity. Tumour assessments were performed every eight weeks for the first 40 weeks and every 12 weeks thereafter.
ROSELLA met both the co-primary endpoints. The patients treated with relacorilant plus nab-paclitaxel had a median survival of 6.5 months compared to 5.5 months in patients treated with nab-paclitaxel alone.
The median OS was 16 months in the relacorilant plus nab-paclitaxel arm and 11.9 months in the nab-paclitaxel arm.
Patients treated with relacorilant plus nab-paclitaxel also had a 35% reduction in the risk of death compared with those receiving nab-paclitaxel monotherapy
In addition, patients in the combination arm had a 30% reduction in the risk of disease progression, as assessed by blinded independent central review, compared with the control arm.
The most frequently observed adverse reactions (including laboratory abnormalities), occurring in more than 20% of patients, were decreased appetite, decreased haemoglobin, diarrhoea, fatigue, nausea, rash, reduced neutrophil counts and thrombocytopenia.