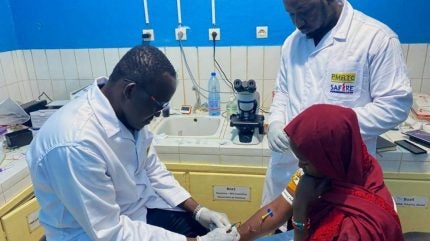
A partnership between Europe and Africa has seen the initiation of a clinical trial of an anti-malaria drug in early pregnancy.
The Phase III adaptive platform trial (NCT06962319), which is part of the SAFIRE project, is funded by the Global Health EDCTP3 and is specifically designed to investigate antimalarials in pregnant women in their first trimester.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The study has already enrolled 13 women in Mali, 31 in Burkina Faso, and eight in Kenya and is comparing the efficacy and safety of two commonly used artemisinin-based combination treatments (ACTs), pyronaridine-artesunate and dihydroartemisinin-piperaquine, against the current standard of care (SoC). Currently, only one ACT, artemether-lumefantrine, is recommended for use in the first trimester.
“This first-of-its-kind trial addresses an important need for the treatment of malaria during the first trimester of pregnancy, being able to treat the infection with safe and effective drugs. The SAFIRE project will provide an evidence base to make additional drugs available for malaria treatment in the first trimester and also contribute to addressing the lack of equity in enrolling pregnant women in clinical trials,” said Dr Henk Schallig, SAFIRE coordinator at Amsterdam University Medical Center (UMC).
The consortium is led by the Amsterdam UMC and also features the involvement of the University of Sciences, Techniques and Technologies of Bamako (USTTB) and the Kenya Medical Research Institute (KEMRI).
The consortium’s study aims to inform national and international treatment guidelines to prevent maternal anaemia, stillbirth, and neonatal mortality, while also reducing the burden of malaria in pregnant women.
Every year, over 12 million pregnant women in sub-Saharan Africa are exposed to malaria, resulting in roughly half a million infections in the first trimester alone. The World Health Organization (WHO) African Region continues to face the most acute crisis, accounting for 94% of all cases and 95% of malaria deaths.
Not only does this pose a challenge for patients, but pregnant women have historically been excluded from clinical research due to safety concerns. This is something the WHO has been calling to change.
Analysis led by the Global Observatory on Health Research and Development of all clinical trials in the International Clinical Trials Registry Platform (ICTRP) reveals that just 4% of clinical trials over the past decade allowed the inclusion of pregnant women. As a result, many pregnant and breastfeeding women are left without treatment options or take prescription medicines off-label, without adequate data to inform safe use.
In a previous conversation with Clinical Trials Arena, Sheila Diamond, director of scientific engagement at Medidata, said this is something that all parties involved in clinical trials need to address to improve access to research for pregnant women.



