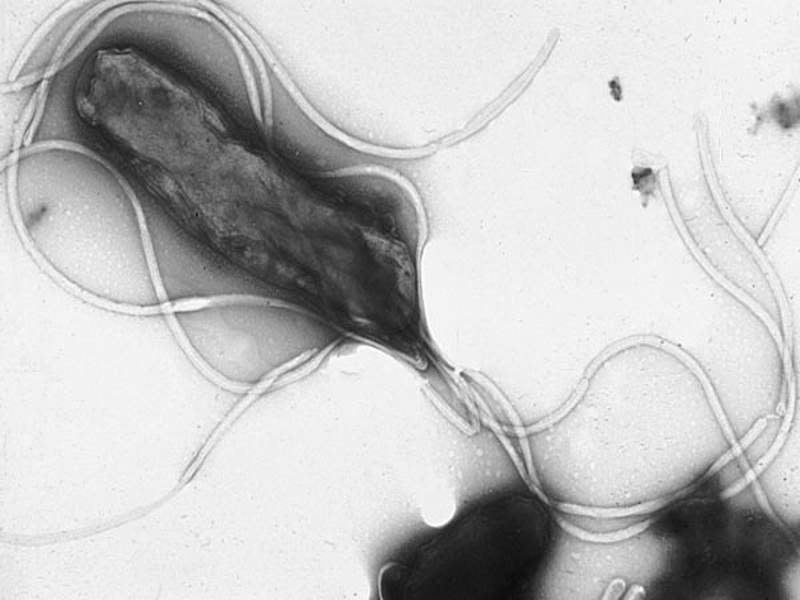

Israeli-based specialist biopharmaceutical firm RedHill Biopharma has commenced a confirmatory Phase III ERADICATE Hp 2 clinical trial of RHB-105 (Talicia) for the treatment of patients with Helicobacter pylori (H. pylori) infection.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Talicia is an oral, fixed-dose combination of two antibiotics and a proton pump inhibitor (PPI) in a single capsule.
The two-arm, randomised, double-blind, active comparator, confirmatory Phase III trial is designed to recruit 444 subjects at approximately 65 clinical centres in the US.
The trial will compare the product with a combination therapy regimen of amoxicillin and omeprazole at equivalent doses in non-investigated dyspepsia patients with a confirmed infection.
Patients will be administered four capsules three times a day of either Talicia or the active comparator over 14 days.
The trial’s primary endpoint is eradication of the infection at 42 through to 70 days after the start of treatment.
The previous Phase III ERADICATE Hp trial of the product is reported to have demonstrated 89.4% efficacy in eradicating the infection when compared to standard-of-care (SoC) treatment.
The firm expects that the potential favourable results from the ERADICATE Hp 2 trial, positive results from the ERADICATE Hp trial and findings from the supportive PK programme will support a US new drug application (NDA) for Talicia.
The US Food and Drug Administration (FDA) has granted the product with qualifying infectious disease product (QIDP) designation, a fast-track development pathway, NDA priority review status and extended market exclusivity in the country for eight years.
Image: Electron micrograph of H. pylori. Photo: courtesy of Yutaka Tsutsumi/Professor department of pathology / Fujita Health University School of Medicine.





