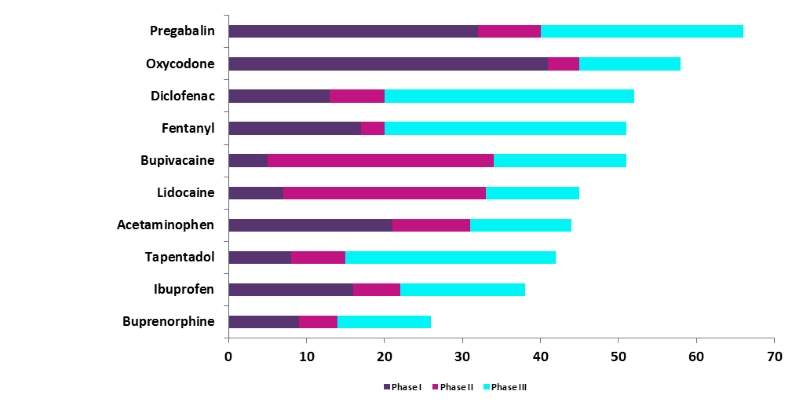
GlobalData undertook analysis of the top ten drugs being investigated within pain clinical trials. Over the ten-year period from 2007 to 2016, Pregabalin was the top drug with the highest number of clinical trials initiated at 66 trials. In second place was Oxycodone with 58 trials, while Diclofenac with 52 trials took third place.
In joint fourth place were Fentanyl and Bupivacaine with 51 trials respectively, and Lidocaine took sixth position with 45 trials. This was followed closely by Acetaminophen with 44 trials.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The final positions were occupied by Tapentadol, Ibuprofen, and Buprenorphine with 42, 38, and 26 trials respectively over the ten-year period.
The phases of these studies were quite balanced across all three levels of development, although Pregabalin and Oxycodone were more concentrated within Phase I and III trials rather than the Phase II stage.
Figure 1: top 10 investigated drugs by phase, 2007-2016

Source: GlobalData, Pharma Intelligence Center, Clinical Trials Database [accessed 29 August 2017]

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataFurther analysis found that Pregabalin has been approved for a number of pain indications, such as diabetic neuropathic pain and general neuropathic pain and it is currently in development for more pain indications such as chronic pancreatitis pain, post-operative pain, polyneuropathy, and peripheral neuropathic pain.
Such an active pipeline is indicative of a broad mechanism of action able to treat a variety of symptoms. This benefits patients affected by pain and enables pharmaceutical companies to penetrate into patient populations where previously there may have been minimal success.



