
As obesity reaches epidemic proportions worldwide, clinical trial teams face mounting pressure to demonstrate meaningful health benefits beyond weight loss alone. With half of adults projected to have a high BMI within the next decade, the challenge for researchers is not only developing effective treatments but also defining endpoints that truly capture patient well-being.[i]
Clinical Outcome Assessments (COAs) and Digital Health Technologies (DHTs) are increasingly central to that mission. Together, they offer a more holistic view of treatment impact – one that considers how patients feel, function, and live their daily lives – and integrating both COAs and DHTs can enhance the design of obesity clinical trials.
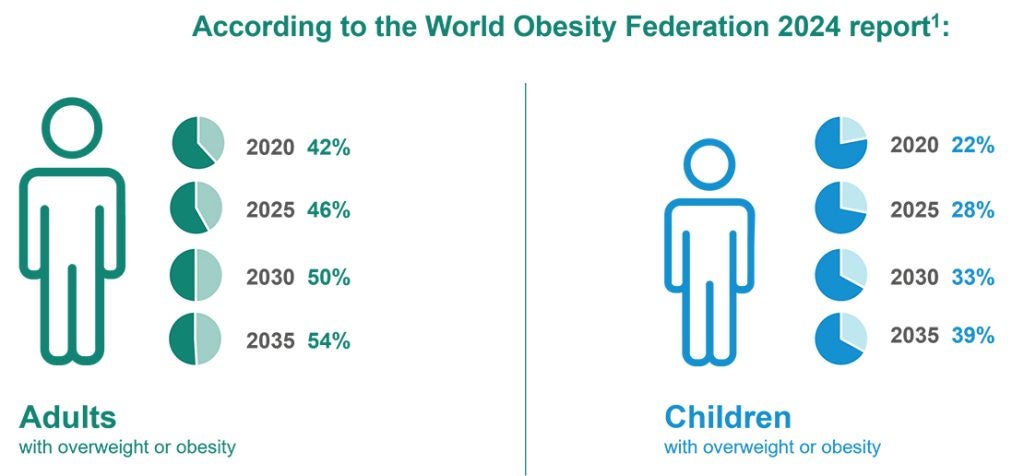
Rethinking endpoints in obesity clinical trials
Traditional obesity endpoints, such as percentage of weight loss or reduction in comorbidities, are no longer sufficient to characterize meaningful outcomes. As the use of GLP-1s and other novel treatments grows, researchers have observed not only fat reduction but also significant muscle loss, which can be detrimental to overall health, despite reducing the number on the scale. This example highlights the limitations of weight-centric endpoints and the need for more comprehensive measures of physical function and mental health.
Over the past decade, obesity trials have begun incorporating more patient-centered outcomes such as quality of life, physical functioning, mental health, sleep, eating behavior or impact on work, as measured by patient-reported outcomes (PROs), like the IWQOL-Lite-CT, the Short Form-36, and the Patient Health Questionnaire-9 (PHQ-9).[ii] Regulatory authorities, including the FDA and EMA, increasingly emphasize the inclusion of PROs in labeling, reinforcing the importance of assessing endpoints that reflect real-world benefit.
The evolving regulatory perspective on COAs
While regulators have long recognized weight loss as the primary endpoint in obesity clinical trials, recent guidance signals a deeper shift in regulatory thinking: the need to contextualize the number on the scale within the broader impact on a patient’s physical and psychological health.[iii] The FDA’s most recent recommendations emphasize this multidimensional approach, encouraging sponsors to assess areas such as physical functioning and neuropsychiatric outcomes (using the PHQ-9 or the Columbia Suicide Severity Rating Scale) alongside traditional biomarkers. This represents a clear regulatory imperative to move beyond descriptive efficacy and build evidence that speaks to meaningful health outcomes. For sponsors, the big opportunity lies in proactively aligning COA selection with both regulatory expectations and the patient experience, ensuring endpoints are credible, interpretable, and relevant to patients.
The patient perspective: defining what matters most
Current regulatory guidance highlights the importance of including patient-reported outcomes (PROs) in obesity clinical trials but does not specify which PROs should be used. To bridge this gap, the Standardise Quality of Life Measurement in Obesity Treatment (S.Q.O.T.) initiative brought together researchers, clinicians, and people living with obesity to define which outcomes truly matter.
The group set out to answer two key questions: What concepts matter most to people with obesity? And Which existing PROs best measure these concepts?
Using systematic reviews, surveys, and global consensus meetings, the group identified 25 key concepts and 17 PROMs that capture the lived experience of obesity. To be included, a PRO had to both capture concepts meaningful to patients and demonstrate strong measurement properties. The findings revealed clear priorities in what patients value most:
- Self-esteem
- Body image
- Energy
- Social participation
These aspects extend beyond the clinic and into daily life. While clinicians tend to prioritize physical and mental health domains, patients consistently emphasize the psychological and social dimensions of their experience.
This patient-led consensus underscores a pivotal evolution in obesity research: clinical efficacy alone is not enough. Trials must also demonstrate meaningfulness and show how interventions improve the aspects of life that patients themselves define as most valuable.
The expanding role of digital endpoints
While regulators have historically emphasized biomarkers and COAs, recent advances show a clear rise in the use of Digital Health Technologies (DHTs) as complementary tools in obesity clinical trials. Wearable sensors, connected scales, blood pressure monitors, and continuous glucose monitors now allow sponsors to capture outcomes such as physical function and metabolic change continuously and in real-world settings.
When integrated thoughtfully with COAs, these digital measures provide a richer, more granular picture of treatment impact. The FDA’s recent guidance affirms that digital endpoints should be fit for purpose and undergo validation consistent with COAs, following frameworks such as V3+. Together, COAs and DHTs enable the selection of tools that are scientifically robust, patient-centered, and meaningful to regulators.[iv]
Toward a holistic view of treatment impact
Leveraging both COAs and DHTs provides a multidimensional view of patient outcomes, combining subjective experience and objective measurement. Often these complementary assessments approach a single concept from varying angles, contextualizing each to provide a more holistic understanding of the patient’s lived experience. Lessons from other therapeutic areas, such as COPD, show how hybrid approaches can capture both the quantity and quality of patient activity, resulting in richer, more interpretable evidence of benefit.
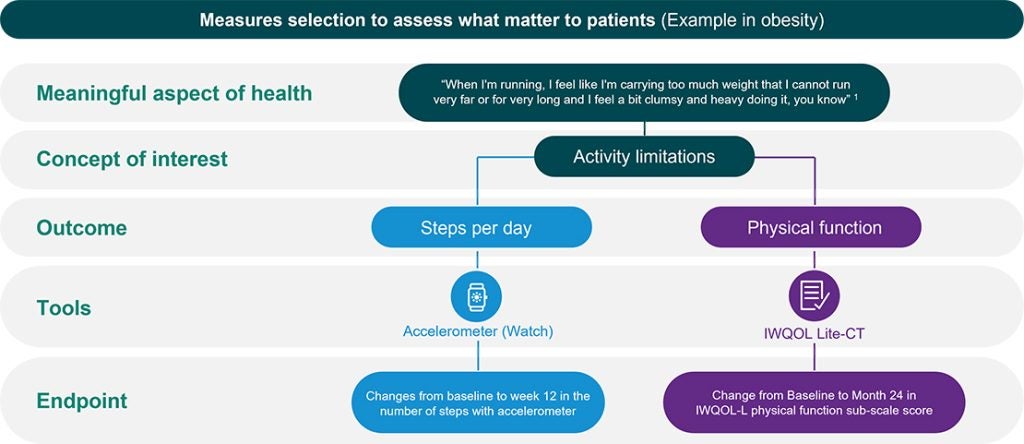
For sponsors working in obesity, this integrated approach can increase the likelihood of regulatory success while aligning trial outcomes with what truly matters to patients.
To learn how to select complementary COAs and DHTs for obesity trials, watch ICON’s recent webinar “Enhancing clinical trials in obesity by integrating COAs and DHTs.”
[i] World Obesity Federation. “World Obesity Atlas 2024.”
[ii] Goetz IA, Sutter C, Abraham T, Kanu C, Boye KS, Symonds T. ”The evaluation of clinical outcomes assessments and digital health technologies in clinical trials for obesity.“ J Patient Rep Outcomes. 2025 Feb 20;9(1):23.
[iii] Food and Drug Administration. Guidance Document: Obesity and Overweight: Developing Drugs and Biological Products for Weight Reduction.”
[iv] U.S. Food and Drug Administration. “Digital Health Technologies for Remote Data Acquisition in Clinical Investigations: Guidance for Industry, Investigators, and Other Stakeholders.” Guidance for Industry



