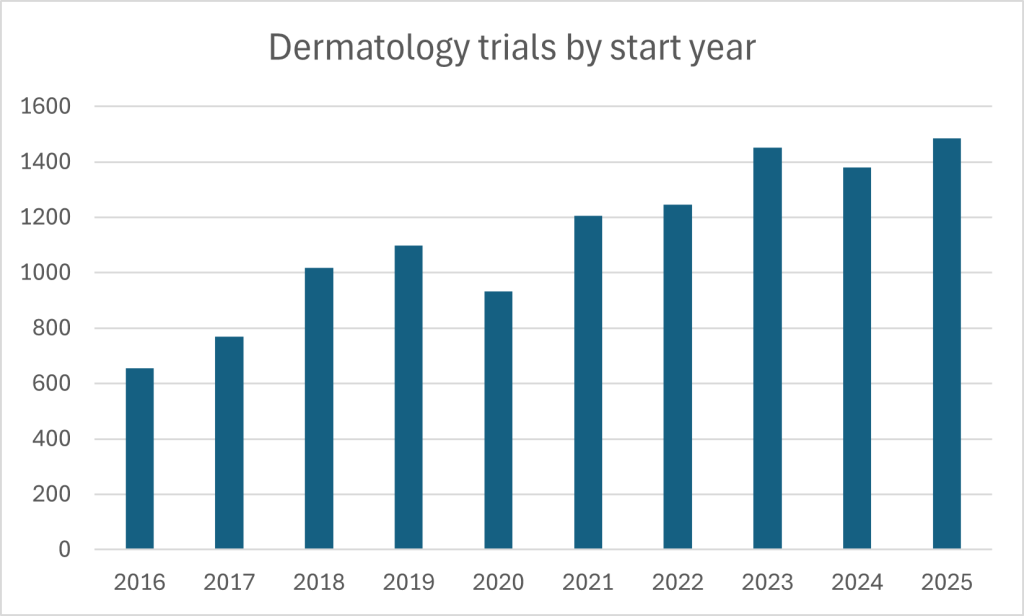
Dermatology – spanning chronic inflammatory disease, infection, wound healing, pigment disorders, hair loss and rare genetic conditions – is nothing if not a broad area of clinical research. That breadth is also contributing to its status as one of the most vibrant fields, both in growth and overall volume, in all of medicine: GlobalData’s clinical trial database suggests more than 16,000 dermatology trials were either completed, ongoing, or planned at the end of 2025. The tempo has quickened. Trial starts have rocketed over the past decade, peaking over the past year.
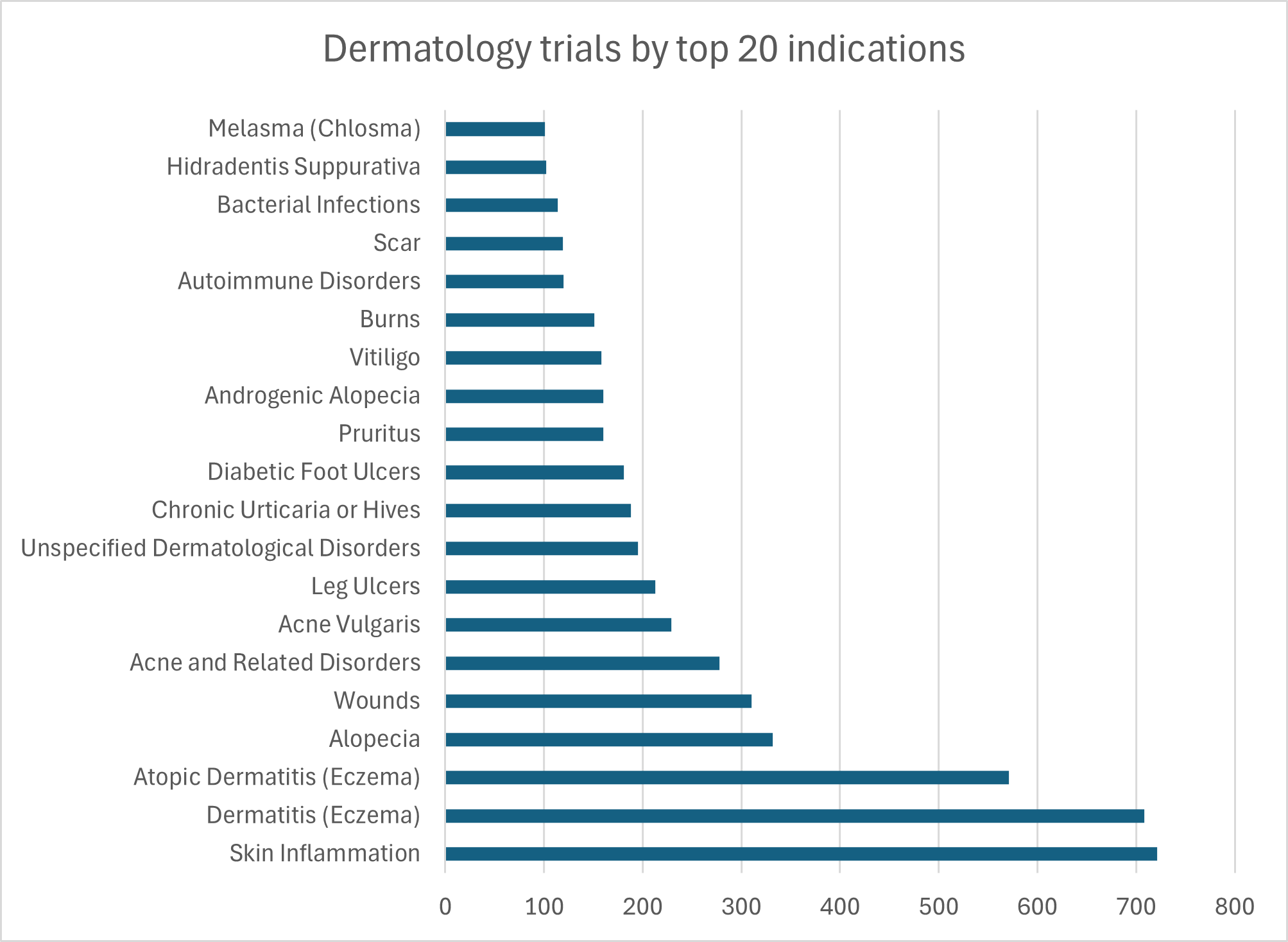
Many programs are moving at the same time and at rapid speed, from small topical studies to complex, multi-country development plans. Geography is shifting too. By December 2025, the Asia-Pacific region had seen a nearly five-fold increase in dermatology trials compared to a decade earlier, and accounted for nearly 8,000 such trials overall, around the same quantity as North America and Europe combined. Sponsors have widened their footprint to match where disease burden is greatest, where investigator networks are deepening and where trial infrastructure has become more capable. For example dermatophytosis, a fungal infection, is expected to reach over 830 million infections by 2029, with the highest prevalence in India and China. Big numbers like that pull research toward the places where patients are, and toward the practical problems that determine whether a trial finishes on time.
Globalization, however, brings challenges. Regulatory requirements differ across the FDA, the EMA and major Asia-Pacific authorities for various drug product types and disease indications.. And dermatology brings its own unique challenges; endpoints are often visual and, therefore, vulnerable to variation in imaging methods, grading standards and assessor training. Dermatological disease may also look different across skin tones, with treatment response and adverse effects varying. Rising therapeutic activity brings with it rising opportunities for research – but those on the frontline must navigate potential pitfalls sensitively.
Major trial trends
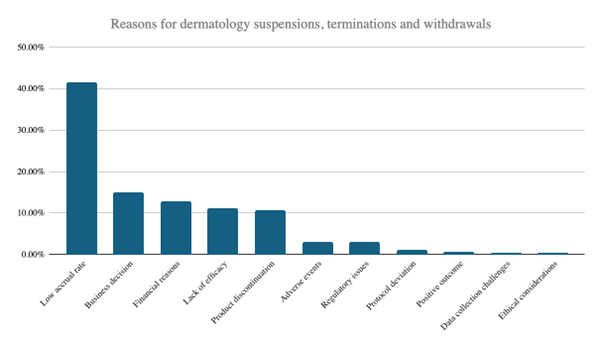
Several currents coursing through today’s dermatology landscape are worthy of scrutiny. One relates to limitations and stumbling blocks in trial capabilities. Visual endpoint assessments – critical to conditions like psoriasis and eczema – require meticulous consistency. Training protocols, whether developed by vendors or key opinion leaders, must ensure that investigators grade lesions uniformly across sites. Most importantly, with low accrual rates the key reason for trial suspensions, termination and withdrawals across dermatology research, sponsors must guarantee seamless flows of vital data if they are to expedite trials. Doing so translates directly into faster, more effective recruitment; failure means the opposite. Licensing and translation work should begin during the proposal process lest it leads to hold ups and compresses enrollment windows. Site start-up likewise benefits from streamlined approaches: this means maintaining libraries of site documents, using contract templates, and waiving qualification visits where prior relationships exist. Once a trial is underway, elements like electronic patient-reported outcome questionnaires must be handled efficiently to avoid them presenting another rate-limiting factor.
Additionally, regulatory divergence remains a persistent obstacle in multinational programs. The FDA’s modern, streamlined pathways, particularly for orphan drugs, can deliver faster approvals than Europe’s more conservative approach to safety endpoints and long-term data. Asia-Pacific agencies vary widely due to multiple standalone authorities across multiple countries, though harmonization efforts are narrowing gaps. Sponsors must anticipate local data requirements early, design trials to enroll sufficient regional subpopulations, and engage regulators before pivotal studies commence.
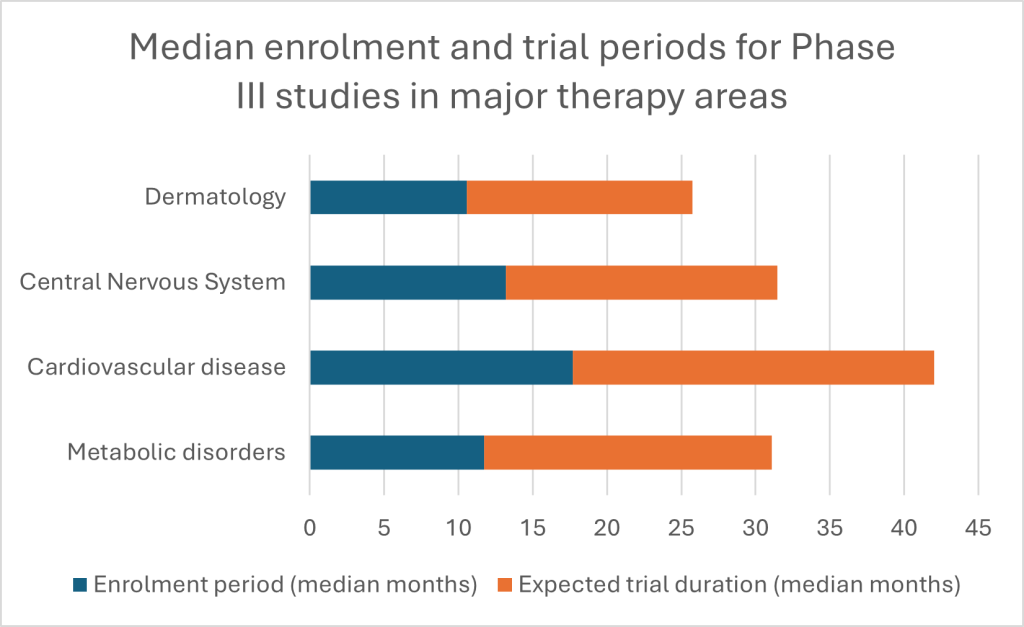
Despite challenges, there are clear reasons to be optimistic. When dermatology trials engage patients efficiently, deploy remote options where appropriate, are consistent with visual endpoints and use focussed data reviews to make monitoring easy, they can move quickly. Median enrolment time for Phase III dermatology trials is around 11 months according to GlobalData estimates, compared with close to one year in metabolic disorders, 13 months for central nervous system studies and 18 months for cardiovascular trials.[1] Total trial periods show a similar advantage versus other major therapy areas. Dermatology, while vulnerable to avoidable bottlenecks, is far from doomed to slow execution; laggards can learn from industry leaders who deploy CRO partnerships and other such research catalysts to turbocharge trial times.
There are other exciting innovations afoot. Consider cell and gene therapies. There are currently 119 such assets in development for 23 dermatological conditions, spanning discovery through pre-registration phases. The pipeline skews heavily toward early stages – 21% in discovery, 39% in preclinical work – but momentum is building. For example, dystrophic epidermolysis bullosa, a rare and currently incurable genetic disorder affecting skin and mucosal tissue, has emerged as a leading indication. It claims the most pipeline assets and the only product currently in pre-registration, alongside five Phase III candidates. The FDA’s approval of Vyjuvek in May 2023 marked a watershed: the first redosable gene therapy for the condition, delivered topically via a modified herpes simplex virus vector. It points the way to new techniques addressing old problems that were once thought intractable across the dermatology landscape.
It is clear that dermatology researchers face a mix of challenges and opportunities over the coming year. How can they most efficiently grapple with the former and lean into the latter?
Staying ahead of the curve in 2026
The pressures of rapid growth, fragmented recruitment, endpoint complexity, global regulation and advanced modalities are pushing sponsors toward partners that can make sense of a changing dermatology market without slowing momentum. Two recent programs illustrate what that looks like in practice.
One recent phase III plaque psoriasis study in adults planned 37 sites for 508 patients. Enrollment finished six weeks early, even after an amendment increased the number of subjects without extending the enrollment period. The final enrolled population rose by 18%, and the program passed a regulatory inspection by China’s drug inspectorate. Speed mattered, but so did quality: the same study faced dual-sponsor requirements and pandemic disruptions that threatened visit schedules and data collection. Collaboration with Caidya, whose global footprint and experience responding deftly to changing protocols, ensured the study was a success.
A second program, a phase II study in moderate-to-severe atopic dermatitis, enrolled 163 subjects in six months across 27 sites, despite the added hurdle of a placebo control. As is the case across the dermatology world, enrollment difficulty represented a looming danger to trial success. Here, execution hinged on practical choices: getting sites ready under restrictions, aligning investigators around recruitment tactics and keeping momentum steady across centers. Again, partnership with Caidya made all this possible and freed the sponsor to focus on the science.
What can researchers learn from these examples as 2026 gets underway? First, recruitment is about incentives, logistics and site selection – there are organizations out there who specialize in all three, and sponsors whose specialty lies in epidemiology should seek them out. Second, dermatology’s reliance on visual assessment makes consistency a core operational problem they should seek to address. Third, global dermatology development will keep expanding in novel regions.
Across all these areas, a CRO can change outcomes – combining local execution with global standards, and by ensuring that measurement, training, documentation, and submission are aligned across regions. Caidya is perfectly placed for that task. Its dermatology experience spans large inflammatory indications and complex multi-site delivery, backed by teams that can support inspection readiness, harmonize endpoint assessment and run studies across the globe. Strategic CROs represent essential partners for the coming year, empowering sponsors to design trials with patients at the center, navigate evolving regulatory landscapes and ultimately deliver more meaningful outcomes in dermatology. Download the whitepaper on this page to learn more.
[1] GlobalData Clinical Trial enrollment analytics database, accessed February 2026.



