As Parkinson’s disease (PD) Awareness Month approaches this April, analysis of the global drug landscape reveals an evolving research focus in PD. While pharmaceutical research has historically concentrated on dopamine signalling, the current pipeline indicates raised interest in alpha synuclein as a therapeutic target.
Parkinson’s disease is a progressive neurological disorder, which is caused by the degeneration of dopamine-producing neurons in the brain. Mainly affecting adults aged over 50 years, symptoms include motor issues such as muscle rigidity and tremors. While no cure exists, current marketed drugs help manage symptoms and improve quality of life.
Most current approved treatments work by modulating and restoring dopamine signalling in the brain. Out of the 64 innovator drugs approved globally for PD, 34 (53%) directly target dopamine receptors, highlighting the continued reliance on dopaminergic mechanisms to manage the disease.
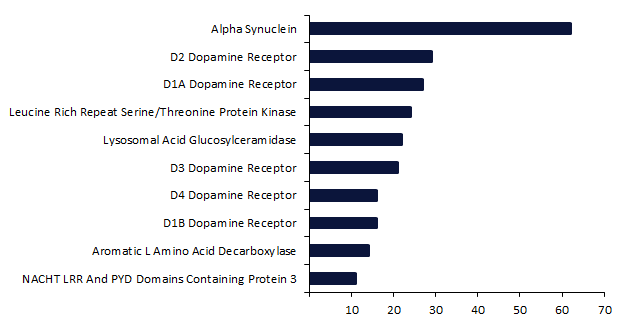
Research activity for PD remains extensive, with 749 innovator drugs in development for their first approval. Dopamine-related mechanisms continue to play a major role in the pipeline, as 50% of the top ten targets are associated with a dopamine receptor subtype, reinforcing the continued relevance of dopaminergic pathways in PD treatment strategies. However, the single most prominent target in the PD pipeline is alpha synuclein, with 62 drugs currently in development, double the 29 pipeline drugs targeting the D2 dopamine receptor, which is the second most popular target (Figure 1).
Figure 1: Most popular targets for pipeline drugs in development for Parkinson’s disease
Alpha synuclein is a presynaptic protein involved in neuronal function. In PD, the protein misfolds and aggregates together, contributing to neuronal degeneration and disease progression. Due to this central role in PD pathology, alpha synuclein has become a key focus for the development of therapies to address the underlying biology of PD and halt disease progression.
Alpha synuclein research remains at a very early stage, as 46 products (75%) are currently found in either the discovery or preclinical phases. This suggests an immature field, as researchers are still translating this biological hypothesis into viable therapies.
Despite this research focus in PD, no alpha synuclein drugs have received regulatory approval for any indication. There are two Phase III PD drugs that target alpha synuclein, hoping to become the landmark approval in this area: Annovia Bio’s small molecule, buntanetap tartrate, which is administered orally, and Roche/Prothena Corp’s intravenously delivered monoclonal antibody, prasinezumab. GlobalData’s Likelihood of Approval tool rates buntanetap tartrate to have a slightly higher chance of approval (24%), compared to prasinezumab (16%). However, the relatively low probabilities of success highlight the scientific uncertainty surrounding alpha synuclein therapies, despite the strong level of research in this area.
Despite growing interest in alpha synuclein as a PD therapeutic target, most programs remain in early development, and the field is yet to deliver its first approval. However, landmark approvals for the late-stage candidates, buntanetap tartrate and prasinezumab, could represent a new era for disease-modifying treatments in PD.