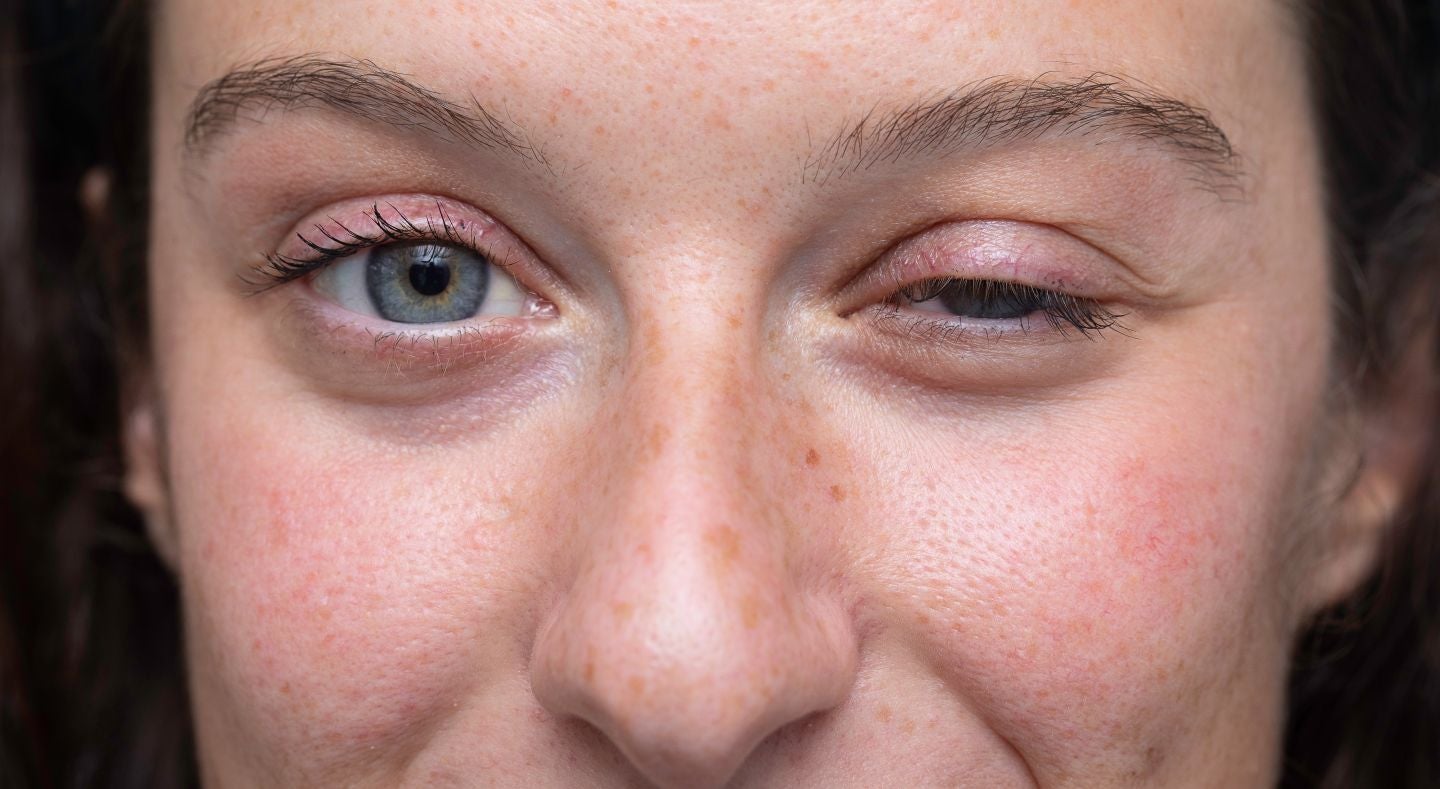
On 26 February, ArgenX announced results from additional analyses of its blockbuster FcRn inhibitor Vyvgart in the Phase III Adapt Oculus study (NCT06558279), evaluating the efficacy and safety of Vyvgart administered by prefilled syringe in adult patients with ocular MG (MGFA Class I) who are acetylcholine receptor antibody–positive (AChR+). The trial met its primary endpoint, which was a change from baseline in Myasthenia Gravis Impairment Index (MGII) (patient-reported outcome [PRO] subcomponent) ocular score at Week 4 (Day 29) compared to placebo.
Vyvgart is a human IgG1 antibody fragment (the Fc portion), which is engineered to bind the neonatal Fc receptor (FcRn) and reduce levels of circulating immunoglobulin G (IgG). Adapt Oculus is a Phase III randomised, double-blinded, placebo-controlled, parallel-group design study evaluating the efficacy and safety of Vyvgart with ocular MG. The study consisted of a part A (approximately 7 weeks) and a part B (up to two years). In Part A, randomised participants (1:1) received four once-weekly injections of Vyvgart or placebo followed by a four-week follow-up. In part B, the open-label extension, participants received two cycles of four once-weekly Vyvgart injections with a four-week interval between cycles. Using patient-reported outcomes on the MGII scale, those in the Vyvgart arm had a mean 4.04-point change versus a mean change of 1.99 in the placebo arm. Patients treated with Vyvgart also saw a reduction of key ocular symptoms—diplopia (double vision) and ptosis (drooping of the upper eyelids).
The results of the trial are exciting. Given that MG is a chronic progressive condition and there is a significant disease burden on patients living with this condition, therapies that can demonstrate early disease control are highly sought after. The Adapt Oculus trial is unique in that it includes a patient population often overlooked in clinical trials, patients presenting with ocular MG. Such patients often rely upon symptomatic therapy, including acetylcholinesterase inhibitors and chronic steroids, and treatments that target underlying pathological mechanisms, such as Vyvgart, are withheld until the patient’s condition worsens. This creates a significant disease burden for MG patients. Key opinion leaders (KOLs) previously interviewed by GlobalData have noted that there is a significant unmet need for effective treatments for patients with ocular MG. These patients are often controlled through symptomatic treatments, and although effective in non-refractory MG patients, refractory MG patients are left with few additional treatment options and are therefore vulnerable to having their MG develop further into generalised MG. The results of the Adapt Oculus trial are a welcome development for AChR+ ocular MG patients, as they indicate that an exciting new treatment option may be on the horizon.
The findings from the Adapt Oculus trial underscore the role of Vyvgart as a transformative treatment for MG. By demonstrating sustained improvements in the MGII scale and a reduction in key ocular MG symptoms, Vyvgart continues to address critical unmet needs in the MG treatment landscape, this time for patients with AChR+ ocular MG. With a proven safety and efficacy profile, Vyvgart continues to display its credentials as a gold-standard disease-modifying treatment in the MG therapeutic landscape. This latest victory in the Adapt Oculus trial paves the way for broader disease management and better patient outcomes in MG. GlobalData estimates that Vyvgart will reach $1.2bn in sales by the end of 2026, prior to biosimilar entry.