GlobalData analysed the number of clinical trials examining the microbiome with start dates between 1 January 2012 and 31 December 2017.
Figure 1 includes clinical trials examining direct treatment by microbiota as well as those trials assessing modulators that influence the internal microbial flora. Additionally, clinical investigations examining therapeutic interventions across a variety of indications where alternations in microbiome composition were assessed as an outcome measure have also been incorporated into the analysis.
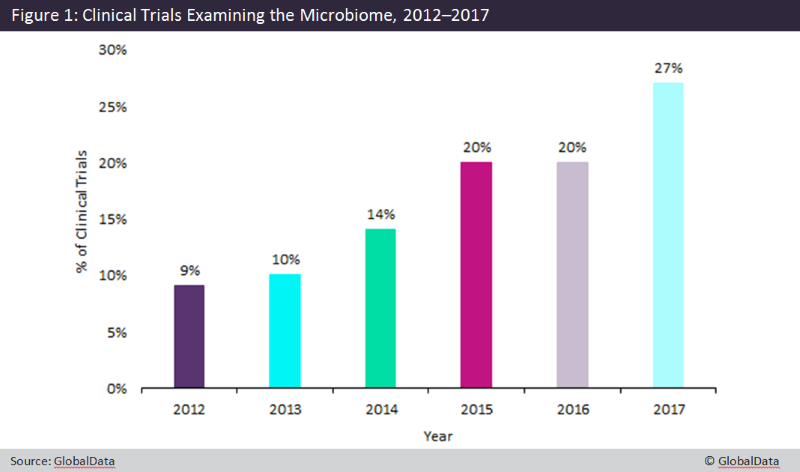
The chart highlights how interest in the microbiome in clinical trials has increased over the past five years, with interest peaking in 2017. Our increased understanding of the human microbiota, namely the gut microbiota and its impact on various health conditions and treatment outcomes, has spurred research efforts where microbiome-based therapeutics are concerned.
The data were clustered into nine main categories based upon frequently examined interventions in clinical trials. The top three categories are prebiotics, probiotics and synbiotics, bacterial transplantations and vitamin/nutritional supplements including dietary interventions, accounting for 36%, 21%, and 15% of clinical trials respectively.
The majority of clinical trials were sponsored by non-industry sponsors that consisted predominantly of academic institutions. Where industry players were concerned, key player Synthetic Biologics had the highest number of clinical trials with interest in microbiome modulation. The company’s planned clinical trials involve the β-lactamase enzyme SYN-004 (ribaxamase) as well as the small molecule SYN-010, a modified-release formulation of lovastatin lactone.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalData



