Trials24 accelerates patient recruitment worldwide to support biopharmaceutical companies and contract research organisations (CROs) in completing their clinical trials on time, bringing innovative drugs to patients faster. We achieve this by providing patient-reported feasibility studies, global digital patient recruitment, and patient materials.
To date, we have randomised more than 3,000 patients globally, contributing between 20% and 65% of all randomised patients and shortening our customers’ clinical trial durations by more than six months on average.
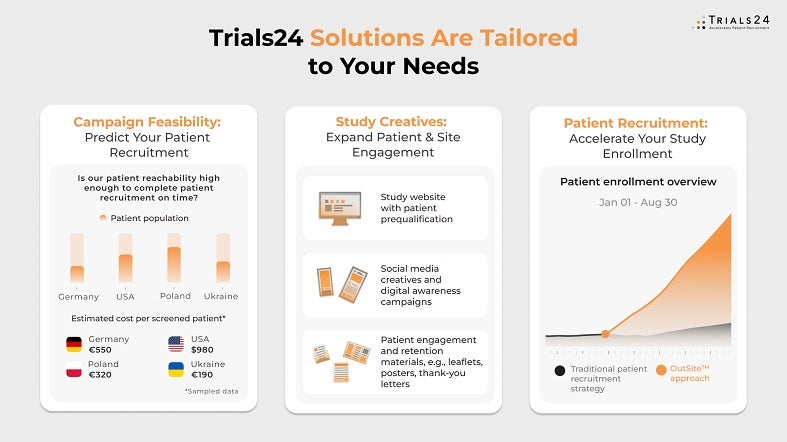
Know our patient recruitment campaign’s ROI before you launch it
Trials24 offers performance prediction through a patient-reported feasibility study before you invest in patient recruitment for clinical trials. We collect real-world data (RWD) from patients based on your study’s protocol, site locations, and countries. With these RWDs, we measure your patient population’s eligibility rate specific to your study protocol and build a recruitment cost model that projects your patient enrolment costs.
This RWD-based scientific approach ensures you know your patient awareness campaign will be ROI-positive before implementing it. Our campaigns typically have an ROI of 7-12x: every dollar invested returns $7-12 in value by significantly shortening your clinical trial duration.
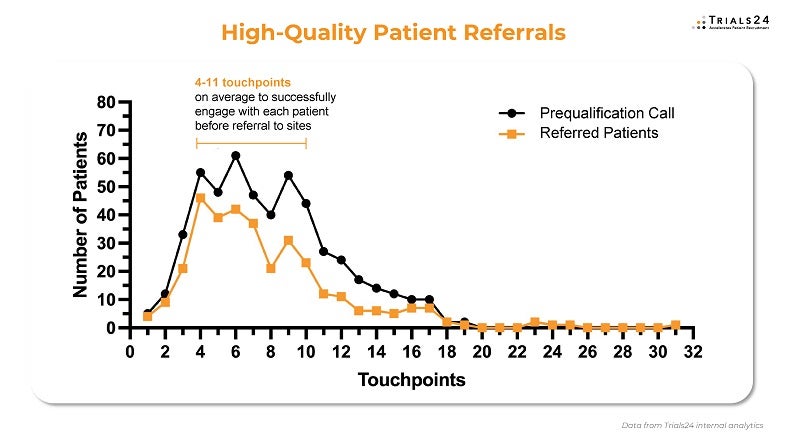
Every patient counts for your study
Our contact centre team contacts patients in under ten minutes and is from the same countries as your patients to ensure cultural and communication fit and the best patient engagement. This way, we achieve inbound call acceptance rates higher than 90% – when patients call us, they reach us immediately. To maintain patient engagement from the first click to enrolment in the trial, we typically have four to 11 touchpoints with the patient, depending on their specific communication needs and personal preferences.
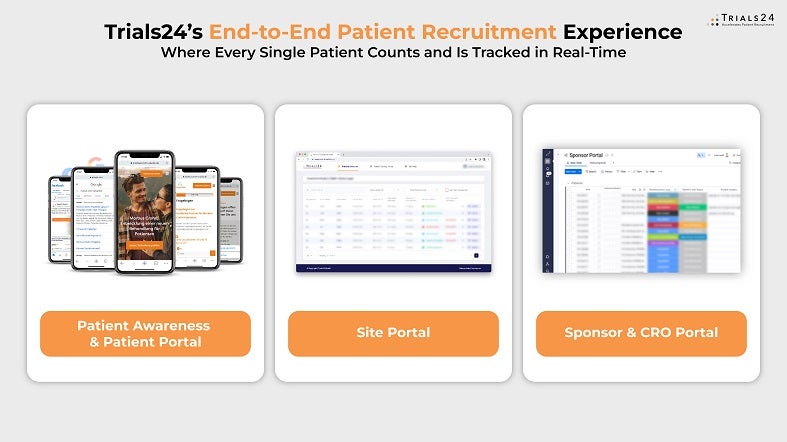
With our Sponsor and CRO Portal’s end-to-end tracking, you can follow every patient along their journey to randomisation in real time – pseudonymised, of course. We ensure that no patient falls through the cracks: every patient counts for your study.
High-quality referrals for your sites
Based on your study’s inclusion and exclusion criteria, we double and triple-prequalify patients so sites have a lower burden and can focus on screening more patients. With our rigorous prequalification process, your sites will enrol up to 40% of our patient referrals.
Selection of our contributions to our customers’ patient recruitment
Inflammatory Bowel Disease (IBD): Phase III Crohn’s disease trial in five countries
- 20-30% of all recruited patients delivered in active countries
- 10-12x Return on Investment (ROI)
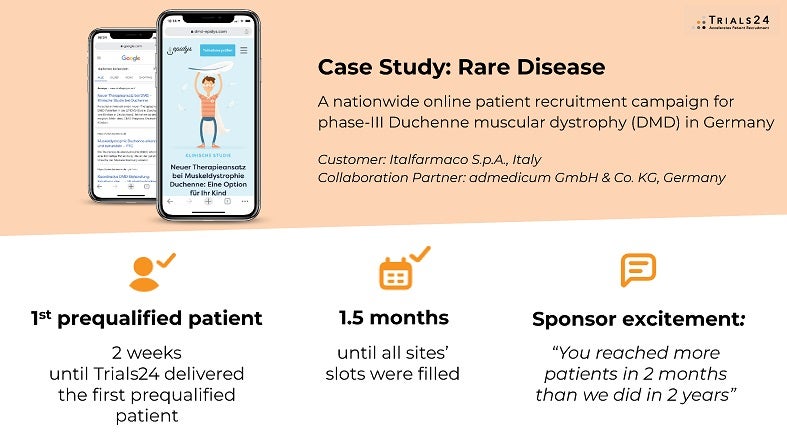
Rare disease: Phase II Duchenne muscular dystrophy trial in Germany
- Reactivated country and filled all screening slots within Germany
- “Trials24 reached more patients in two months than our CRO and sites did in two years” – feedback from our customer
Infectious disease: Phase II Herpes labialis trial in Germany
- Projected end of recruitment a year in advance with achieved accuracy of ± one week
- Delivered 65% of all patients and shortened study duration by 18 months
Rare cancer: Phase I AML, MDS, MM (second and third-line patients) trial in Germany
- Ensured the study was top-of-mind at the sites due to disease- and target-group-specific, ongoing patient awareness campaign
- Contributed 25% of all screenings
Sexually transmitted disease: Phase II Herpes genitalis trial in Germany
- Adjusted the study to market dynamics of the Covid-19 pandemic and redeemed a three-month recruitment pause
- Delivered 60% of all patients and shortened study duration by 12 months
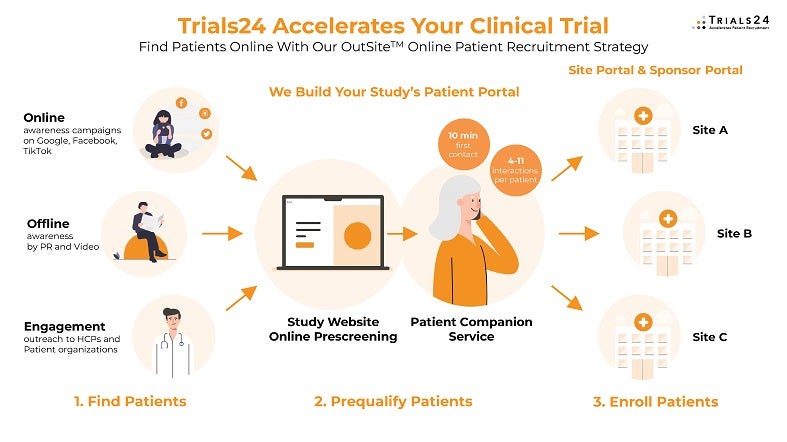
Optimised patient recruitment by finding patients your sites can’t reach
How does this work for your study? When your clinical trial patient recruitment is delayed, your sites are not delivering the patients as initially projected. You can address this by expanding the number of sites, which can take months. Alternatively, for a prompt result, you can seek our assistance in finding patients your sites don’t have access to.
Trials24 accelerates patient recruitment for clinical trials, including hybrid and decentralised trials, by finding patients online in places that most sites can’t access. By reaching patients on Google, Facebook, Instagram, or TikTok, we tap into a patient pool that your sites may not be able to reach. By bringing these patients in from outside your sites – a method we call the ‘OutSite™ approach’ – we complement any site-based recruitment strategy.
Trials24 solutions to predict and accelerate patient recruitment and patient retention
Trials24’s patient-reported feasibility study collects real-world data (RWD) based on your study protocol to predict enrolment costs, ensuring our campaign works before you invest in it. By assessing the eligibility rate of the patient population, we develop a recruitment cost model that anticipates patient enrolment expenses.
Our online patient recruitment services find patients based on online awareness campaigns using search engines and social media platforms, namely Google, Facebook, Instagram, and TikTok. We prequalify and enroll these patients through our double and triple prequalification process with online pre-screeners and phone interviews by our patient contact centre, which responds to any inbound patient contact within ten minutes.
Trials24 designs creative materials for patients and sites to raise awareness for your clinical trial and increase patient retention. We ensure timely delivery with our in-house medical writing capabilities and translation services into more than 30 languages.
About Trials24
Founded in 2017 in Germany, Trials24 operates globally, with its main headquarters in Munich. We help our customers achieve their patient enrolment goals by providing patient-reported feasibility studies to predict recruitment costs, online patient recruitment campaigns to accelerate enrolment, and study creatives to increase patient retention.
Our services have gathered the confidence of clients ranging from small biotechnology firms to top ten pharmaceutical companies. We consistently deliver 20-65% of all randomised patients and can shorten the study recruitment period by six months or more. If you want to learn more about how we may help accelerate your clinical trial patient recruitment, please contact us at info@trials24.com.