According to the American Lung Association, almost 400,000 people die from lung disease each year in the US. The British Lung Foundation puts rates of infection at one in every eight people, approximating to eight million suffers, resulting in death rates of close to 150,000 people a year.
Worldwide, lung disease is the second-biggest killer after cardiovascular disease, and according to lung foundation figures, of 68 million deaths predicted in 2020, 11.9 million will die from respiratory diseases such as asthma, allergic rhinitis, pneumonia, chronic obstructive pulmonary disease (COPD) and bronchitis.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The development of drugs to treat such disorders has stepped up in recent decades, with the US leading the charge. Within different diseases, however, clinical trial rates differ as does investment. Using data from 70 registries across the globe, taken from 1982 to January 2011 GlobalData’s “Respiration Disorders Global Clinical Trials Review, Q1, 2011” provides compelling insight into the state of the industry.
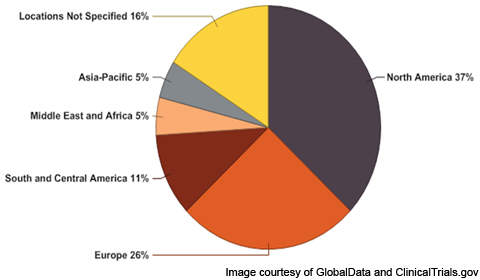
North America leads the pack
In total there were 19 clinical trials conducted on respiration disorders, as of January 2011, of these, seven clinical trials were in North America. More than 35% of clinical trials are conducted in North America followed by 26% in Europe. Within North America, the US has the highest number of respiration disorders clinical trials.
Among the European countries, Norway has the highest number of respiration disorders clinical trials followed by Sweden, Germany and Denmark, as of January 2011. In the Asia-Pacific region, as of January 2011, India has the highest number of respiration disorders clinical trials.
Majority of the respiration disorders clinical trials are in progress
As on January 2011, there were 11 clinical trials in progress and seven trials completed. Only one trial was marked as terminated / suspended or withdrawn due to the lack of efficacy, safety and accrual of subjects. Success rates of the trials were based on completed versus uncompleted (terminated, suspended or withdrawn) trials.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataThe number of respiration disorders clinical trials conducted decreased by 33% for the period 2005-2010. The average number of patients recruited was highest in the year 2009.
Prominent sponsors of respiration disorders clinical trials
In the last decade, Pneumedicare; Oslo University Hospital Health; National Institutes of Health Clinical Center; National Heart, Lung, and Blood Institute; Mayo Clinic; Hospital del Mar; Hospital de Clinicas de Porto Alegre; Department of Health; Dainippon Sumitomo Pharma Co and Cretan Critical Care Society conducted highest number of clinical trials.
In the year 2011, Pneumedicare, Oslo University Hospital Health and National Institutes of Health Clinical Center have the highest number of respiration disorders clinical trials.
Entry of generics to erode the allergic rhinitis market
The allergic rhinitis market was valued at $5.8bn in 2009. The market increased from $4.1bn in 2001 to $5.8bn in 2009 at a compound annual growth rate (CAGR) rate of 4.5%. The market is expected to erode slightly in the forecast period. The erosion of the market is due to the entry of new generics and cost effective branded drugs. The market comprised ten approved drugs in 2009, increasing from three in 2001 to ten in 2009. In the past, an increase in prevalence and a high treatment-seeking rate attracted many players to the allergic rhinitis market. Although the incidence and prevalence rates are increasing, falling drug prices will erode the market at a rate of 0.5% (CAGR) from 2009 to 2017.
Meda, GlaxoSmithKline (GSK), Merck & Co, and AstraZeneca are the leading competitors in the global allergic rhinitis market. The competitive landscape is highly populated with ten products, six of which can be described as strong. This market is characterised by high levels of self medication.
Asthma market is forecast to show marginal growth
The global asthma market is estimated to be valued at $12.4bn in 2009. It is expected to grow at a CAGR of approximately 1.5% from $12.4bn in 2009 to $14bn by 2017. Although the respiratory market has expanded considerably from 2004 to 2009, growth in the seven major markets is expected to slow down from 2010 to 2012.The decline is sales during this period will be due to patent expiries of leading brands. Growth is driven by the expansion of sales in existing classes, the launch of major new products with safety and convenience advantages like route of administration, once-daily administration, and combination therapies and so on.
Symbicort, Advair and Singulair will be the leading competitors of the global asthma market. GlaxosmithKline, AstraZeneca and Merck will continue to be the market leaders until the patents on their drugs expire in between 2010-2012.
Pneumonia therapeutics market forecast to decline
The global pneumonia market was worth $1.6bn in 2009. It is forecast to decline at a CAGR of 6% for the next seven years to reach $940m by 2017. The projected decline rate is primarily attributed to the patent expiries of Levaquin, Avelox and Zyvox by the end of the forecast period. However, increase in the treatment seeking and diagnosed population, and the ever-increasing development of bacterial resistance to currently marketed antibiotics is expected to sustain the growth in the pneumonia market.
The current competitive landscape for pneumonia drugs is strong. There are several marketed products for the treatment of mild to severe pneumonia, which include cephalosporins, macrolides, carbapenems, penicillins, fluoroquinolones, tetracyclines and miscellaneous antibiotics. All treatments are based on attempts to clinically cure and eradicate the pathogens. The monotherapy regimens and combination therapies have satisfactory efficacy and safety profiles. However, the patent expiries of major prescribed cephalosporins and Zyvox during the forecast period would lead to the pneumonia market being increasingly genericised.
Positive outlook for COPD market
GlobalData estimated the COPD market to be worth $8.4bn in 2009 and projects it to grow at a CAGR of 4.7% to reach $12.2bn in 2017. The development of novel once-daily inhaled therapies is expected to provide new treatment options for COPD patients worldwide, and will play a major role in the expansion of the COPD market in the coming years. A steady increase in the incidence rate, the smoking population and in injurious environmental factors in most countries has caused the COPD market to grow significantly.
Bronchitis Market estimated to decline by 3.5%
The global bronchitis market was valued at $459m in 2009 and is forecast to decline by 3.5% annually for the next seven years, to reach $344m in 2017. The typical form of bronchitis is self-limited. The bronchitis market is characterised by many generics and over-the-counter (OTC) drugs. NSAIDs (non-steroidal anti-inflammatory drugs), cough suppressants and antipyretics are merely supportive therapies. Antibiotics are highly prescribed to treat the atypical form of the disease. The decrease in the market size is attributed to the patent expiration of the branded drugs and the uptake of generic drugs.
For further details, please click here.
GlobalData is an industry analysis specialist company providing business information products and services. Its highly qualified team of analysts, researchers, and solution consultants use proprietary data sources, and various tools and techniques to gather, analyse and represent the latest and the most reliable information essential for businesses to sustain a competitive edge.





