Pharma Technology Focus is free for the iPad. Download our app to read the latest issue and browse our back issues for free.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Researchers at Ghent University in Belgium have found a promising new avenue into asthma treatment after discovering that contact with farm dust causes the body to produce a protein which makes the respiratory tract react less severely to allergens. We speak to the researchers to find out more about these findings, which could mark a step forward in the development of preventative treatments for asthma, and the road ahead.
We also hear from industry consultants about the ten key actions required by manufacturers to meet the EU-FMD Safety Features requirements, and how companies can use the new IDMP implementation deadlines to refine their strategies. Plus, we get an insight into the market for dry eye syndrome treatments from GlobalData, find out shy HSDD drug Addyi remains controversial despite its FDA approval, and ask whether clinical trials could be designed in a way that reduces the use of placebos.
Read the issue for free on your iPad through our app, or if you’re on a desktop computer you can also read it in our web viewer.
In this issue
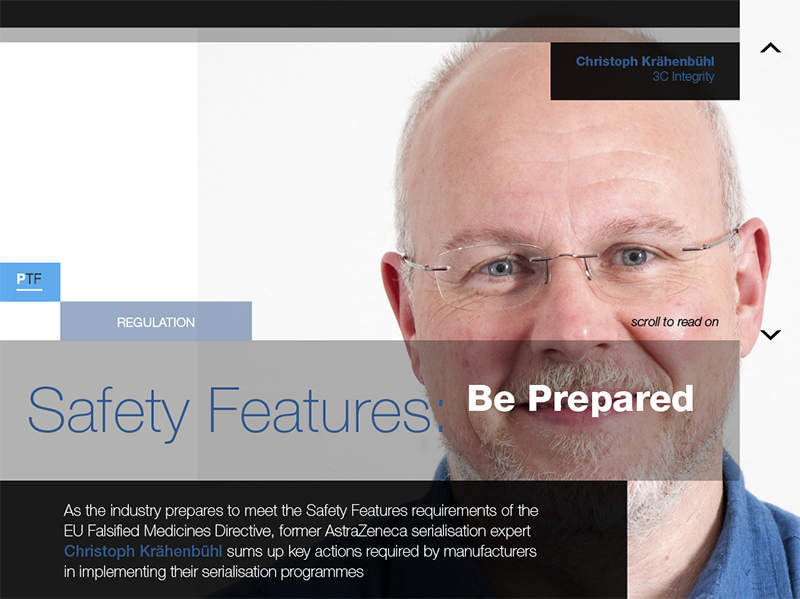
Safety Features: Be Prepared
As the industry prepares to meet the Safety Features requirements of the EU Falsified Medicines Directive, former AstraZeneca serialisation expert Christoph Krähenbühl sums up key actions required by manufacturers in implementing their serialisation programmes.
Read the article.
IDMP: Time to Plan Ahead
The new, phased plan for implementation of the IDMP standards offers the life sciences industry valuable breathing space, allowing it to refine strategies and assign resources, but it shouldn’t be seen as an excuse to put off action, says Sophie Daniel of ProductLife Group.
Read the article.
All Eyes on Shire
GlobalData’s senior analyst for neurology and ophthalmology Catherine Daly, PhD, discusses recent events in the dry eye syndrome treatment market and evaluates the potential impact of recent news for Shire’s promising drug lifitegrast.
Read the article.
Not a Wonder Pill
Earlier this year the FDA approved the first drug for sexual desire disorder in women, but do the drug’s modest benefits outweigh the risks? Elly Earls investigates.
Read the article.
Answers From Dust
Children growing up on farms have a far lower risk of developing asthma and other respiratory allergies, and recently researchers found out why. Abi Millar asks how the discovery could pave the way for a vaccine against asthma.
Read the article.
Finding a Better Way
Fear about receiving the placebo is one of the biggest barriers to clinical trial recruitment. Elly Earls takes a look at new trial set-ups that aim to reduce the number of patients in the placebo group, and the progress that has been made.
Read the article.
Next issue preview
The past year has seen an increased focus on safety as well as continued efforts to make processes more efficient and transparent, with regulatory authorities around the world focusing on streamlining data and improving the consistency of information. We round up the most significant regulatory developments of 2015 and ask how they will impact the life sciences sector in the coming year.
We also take a look at the Nobel prize-winning research behind the discovery of avermectin, and find out how the development of the world’s first 3D stem cell printer could pave the way for personalised medicines as well as reducing the industry’s reliance on animal testing. Plus, we investigate the growing divide between big pharma and healthcare practitioners in the UK, and ask how the relationship between the two sides can be improved for the benefit of patients.
Digital magazine FAQ
You can read Pharma Technology Focus for free on the iPad. Download our app from the App Store to read the latest issue and browse the back issues in our archive. Sign up for a free subscription in the app and never miss a new issue.
You can also continue to read the desktop version for free on our web viewer. (Browser compatibility: The web viewer works in the latest two version of Chrome, Firefox and Safari, as well as in Internet Explorer 9 and 10. Some features may not be compatible with older browser versions.)





