Abivax has reported positive topline results from its Phase lla ABX464-101 clinical trial evaluating the safety and efficacy of ABX464 for the treatment of ulcerative colitis (UC).
The randomised, double-blind, placebo-controlled trial enrolled 32 patients with moderate-to-severe active UC who did not respond to immunomodulators, anti-TNFα, vedolizumab and/or corticosteroids.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
As part of the trial, 29 of the total enrolled patients were randomised in 2:1 ratio to receive a once-daily oral dose of ABX464 at 50mg or placebo for two months.
The trial was carried out at 15 centres in six European countries, including Belgium, France, Germany, Austria, Hungary and Poland.
The final eight-week top-line results from this trial showed that ABX464 achieved 35% of clinical remission compared to the 11% achieved by placebo.
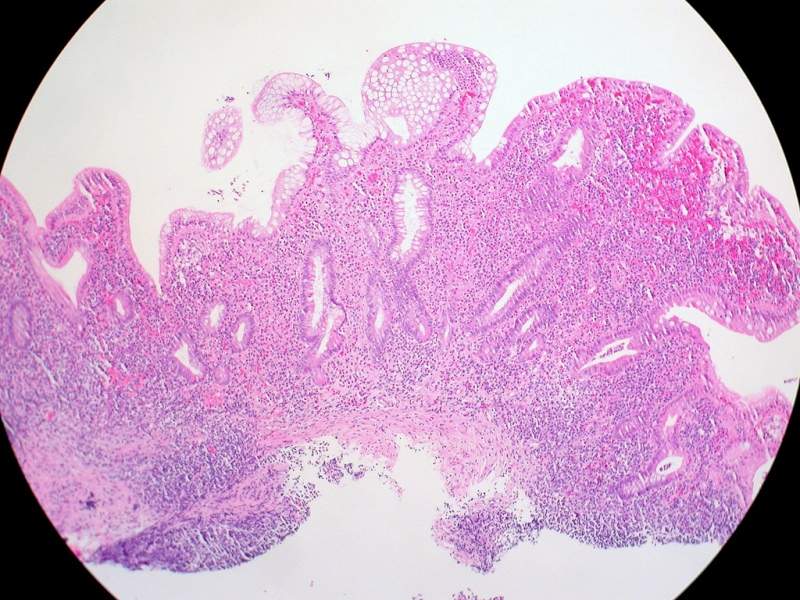
Additionally, 50% of patients treated with ABX464 experienced colorectal mucosal healing as against 11% of the patients who received placebo.
The trial also found that 70% of patients who received ABX464 demonstrated clinical response, compared to 30% of patients treated with placebo.
The data suggested that ABX464 was safe, well-tolerated, and also showed a statistically significant efficacy based on both clinical and endoscopic endpoints of the trial.
ABX464-101 trial principal investigator Dr Severine Vermeire said: “Even with the introduction of biologic treatments in recent years, there is still a large unmet medical need in ulcerative colitis, as too many patients do not respond or stop responding to treatment.
“This well-conducted clinical study provides evidence of a robust and consistent efficacy signal of ABX464 across all clinical and endoscopic endpoints, as well as on biomarkers evaluated.
“These results are very promising and we fully endorse the further development of this exciting new oral compound, both in ulcerative colitis as well as in other inflammatory diseases including Crohn’s disease.”
Furthermore, in four countries where the ABX464-101 study is occurring, patients completing the trial had the option to cross over into a 12-month open-label follow-up study, ABX464-102.





