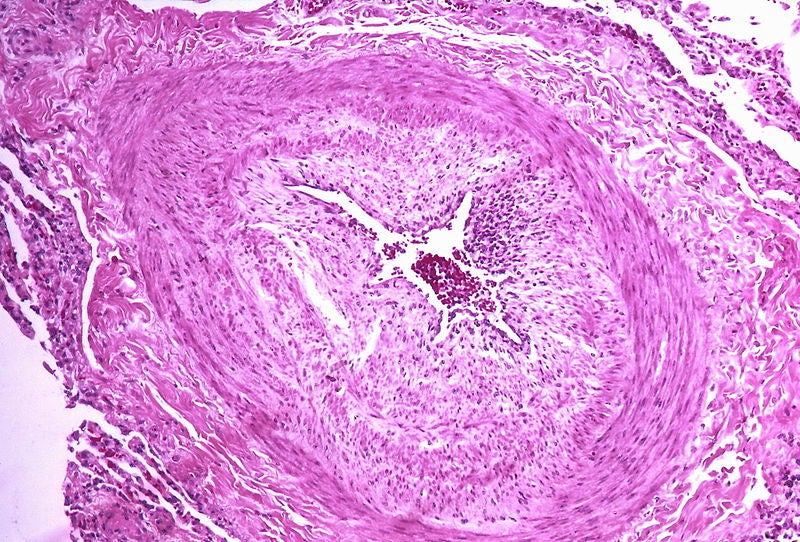
Acceleron Pharma has completed enrolment for the PULSAR Phase II trial of sotatercept to treat patients with pulmonary arterial hypertension (PAH).
The PULSAR trial has enrolled a total of 100 patients with PAH, a rare and chronic disorder caused due to the contraction of small pulmonary arteries and elevated blood pressure in the pulmonary circulation.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Additional patients who are currently undergoing screening will be eligible to be randomised into the trial over the next few weeks.
Acceleron is also enrolling patients with PAH in the Phase II SPECTRA exploratory trial of sotatercept.
Top-line results of the trial are expected during the first quarter of next year.
Acceleron Pharma Pulmonary Medical Research vice-president Janethe de Oliveira Pena said: “We’re thrilled with PULSAR’s rapid enrolment over the past 12 months, which underscores the urgency for new therapeutic options for patients with PAH.
“We believe that through its targeting of underlying disease mechanisms, sotatercept, when combined with standard-of-care therapies, has the potential to alter the PAH treatment landscape.”
The randomised, double-blind, placebo-controlled PULSAR Phase II trial has been designed to evaluate the efficacy and safety of sotatercept in PAH patients.
The trial’s primary endpoint is the change from baseline in pulmonary vascular resistance (PVR) over a treatment period of 24 weeks.
Acceleron noted that the key secondary endpoint is change from baseline in six-minute walk distance.
A total of at least 100 patients will be randomised in a 3:3:4 ratio. They will be given placebo, sotatercept 0.3 mg/kg, or sotatercept 0.7 mg/kg subcutaneously every 21 days with standard-of-care therapies in combination.
The trial participants will be eligible to continue in the extension period for 18 months, following the six-month double-blind treatment period.
Sotatercept, an investigational therapy that is not approved for use in any country, is currently being evaluated in the PULSAR and SPECTRA Phase II trials in PAH.





