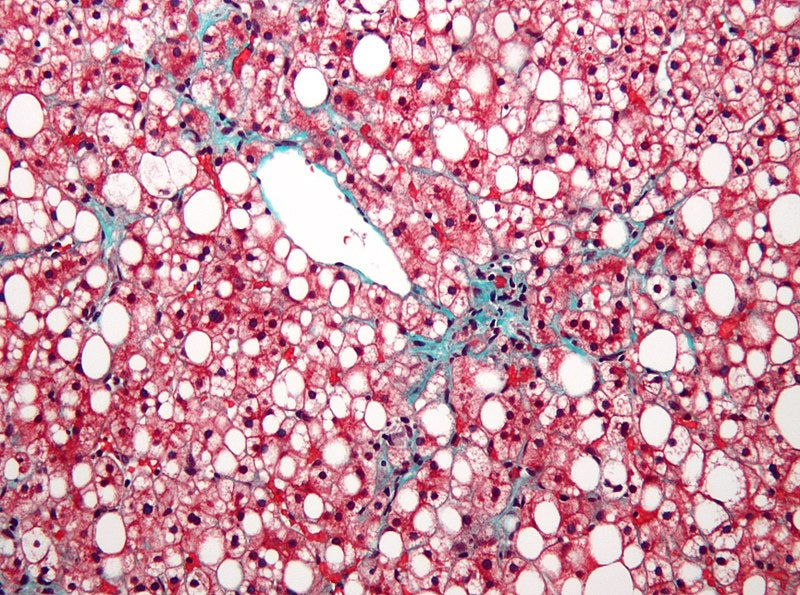
Akero Therapeutics is set to expand the ongoing Phase IIa BALANCED study of Fc-FGF21 fusion protein AKR-001 to include an additional cohort of subjects with non-alcoholic steatohepatitis (NASH) who have compensated cirrhosis (F4), Child-Pugh Class A.
AKR-001 can mimic the biological activity profile of endogenous hormone FGF21, which regulates lipid and energy metabolism, and is secreted throughout the body to reduce cellular stress.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
A total of 30 patients with NASH, demonstrated at baseline by liver biopsy to have cirrhosis with a fibrosis score of four, will be randomised 2:1 to receive either 50mg of AKR-001 or placebo for a period of 16 weeks.
The expansion cohort’s primary objective is to evaluate the safety and tolerability of treatment with AKR-001 in NASH patients at the greatest risk of progressing to end-stage liver disease.
The 50mg dose for this cohort has been selected based on the modelling of data from the Phase Ib trial in Type 2 diabetes, as well as the availability of drug product.
Akero Therapeutics chief development officer Kitty Yale said: “Cirrhotic patients with stage 4 fibrosis present a significant unmet medical need in NASH because of the risk of decompensation and liver failure, where liver transplant becomes the only treatment option.
“AKR-001 may be able to help this vulnerable population.”
NASH, which is closely linked to obesity and diabetes epidemics, is a serious form of non-alcoholic fatty liver disease (NAFLD) and is estimated to affect 17 million Americans.
Based on observations from clinical trials of AKR-001 and other FGF analogues, it has been found that the protein has the potential to reduce liver fat, cellular stress, inflammation and fibrosis in people with NASH.
Last July, Akero announced the dosing of the first patient in the Phase IIa trial of AKR-001.





