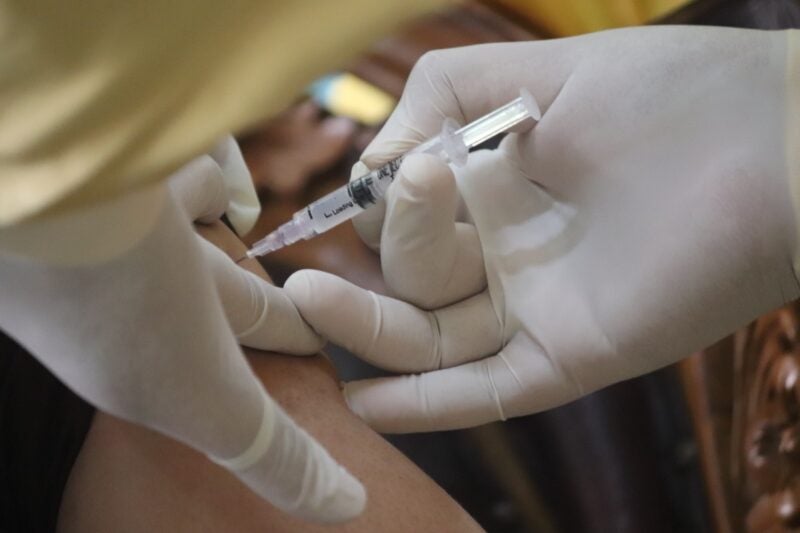
Akston Biosciences has reported positive top-line data from its Phase II clinical trial of AKS-452, a ‘universal’ booster vaccine for Covid-19.
Carried out and managed by contract research organisation TRACER Europe, the open-label, non-randomised, safety and efficacy trial enrolled 70 healthy participants.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
These subjects were aged between 18 and 85 years and were given a 90µg dose of the AKS-452 antigen.
According to the interim findings, the vaccine was found to be well tolerated, with a favourable safety profile in line with registered shots.
In addition, 93% of participants who had received vaccines of Pfizer, Moderna, Johnson & Johnson (Janssen), and AstraZeneca showed a rise in neutralising antibody titers on receiving a single AKS-452 dose.
At day 28, the average neutralisation titers in trial subjects rose four- and five-fold against the SARS-CoV-2 virus’ Wuhan strain and Omicron variants, respectively.
No safety concerns were observed in the trial.
A second-generation vaccine, AKS-452 is based on the new Fc-fusion platform of Akston.
Akston Biosciences president and CEO Todd Zion said: “The favourable safety and immunogenicity profiles at the interim analysis of this study, particularly against variants, support a Phase III universal booster study for AKS-452.
“Boosting immunity in vulnerable populations will be needed for years to come and vaccines must be low cost and easy to distribute.”
In May this year, the company dosed the first set of subjects in the Phase II trial of AKS-452 as a booster.
A Phase II/III trial of the vaccine given as a two-dose regimen in 1,600 participants was previously concluded in India.
The positive trial data was submitted as part of an Emergency Use Application in the country.





