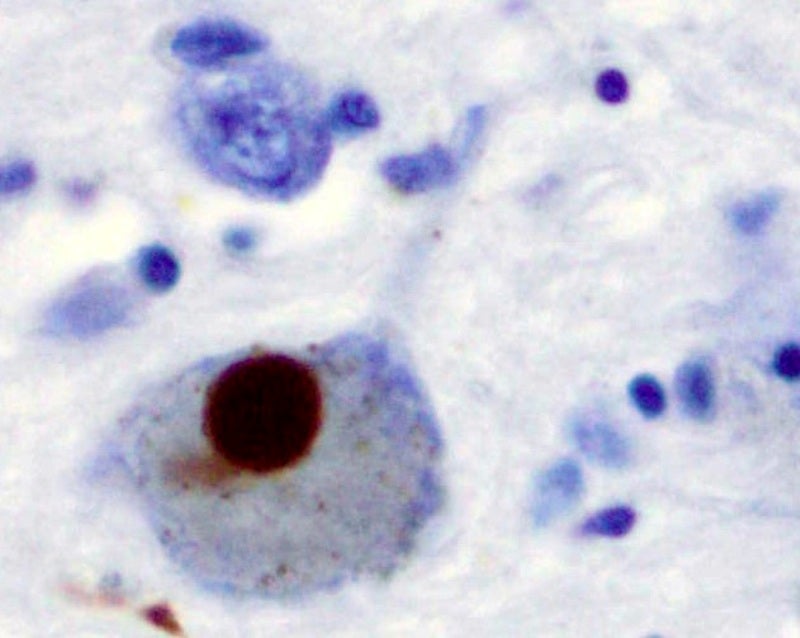
Biotechnology firm Alkahest has started dosing patients in the Phase II AKST4290-211 (TEAL) clinical trial to treat patients suffering from Parkinson’s disease.
AKST4290 is an oral inhibitor of C-C chemokine receptor type 3 (CCR3). The drug is designed to block the activity of an immunomodulatory protein called eotaxin, which is associated with certain age-related diseases.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The trial is part-funded by The Michael J Fox Foundation for Parkinson’s Research (MJFF).
The drug’s action on eotaxin is expected to decrease the inflammation of Parkinson’s and other age-related diseases.
AKST4290-211 is a double-blind, placebo-controlled trial to assess the effect of AKST4290 on motor function and daily activities in 120 patients.
Participants will be treated with 400mg AKST4290 or placebo twice daily. The trial involves a 12-week treatment period and 30 days of follow-up.
The primary endpoint is the motor function in off-medication state at week 12, defined using the Movement Disorder Society’s Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) Part 3.
The study will also track secondary and exploratory endpoints, including safety and tolerability, along with other clinical and functional endpoints in the on-medication state.
Alkahest CEO and chairman Karoly Nikolich said: “Evidence suggests that the immune system plays an important role in the development and progression of Parkinson’s disease, a disease which impacts nearly one million Americans.
“AKST4290 suppresses immune-mediated pathways and chronic inflammation and, if effective, could represent an important step forward in the treatment of Parkinson’s disease.”
Apart from Parkinson’s, AKST4290 is being studied in Phase II trials for the treatment of neovascular age-related macular degeneration.
In December last year, Alkahest commenced a Phase II study of GRF6021 to treat Parkinson’s associated with mild cognitive impairment or dementia. GRF6021 is a human plasma fraction developed by the company in collaboration with Grifols.



