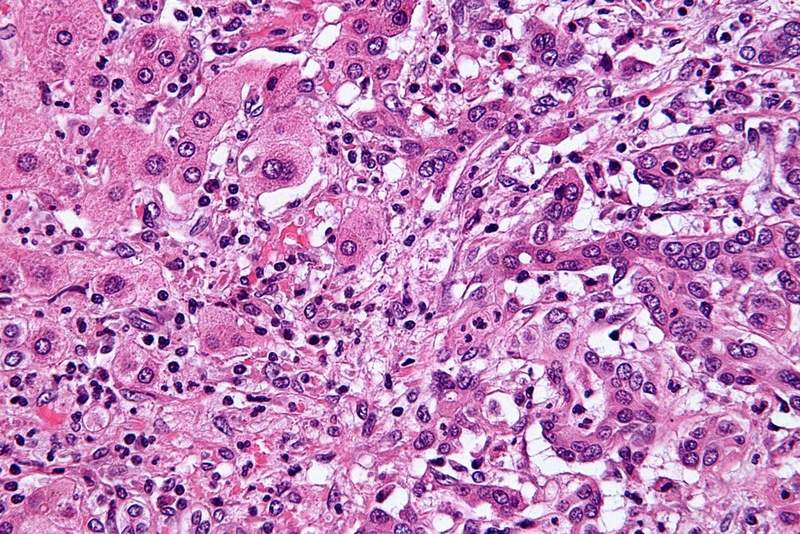
US-based biopharmaceutical firm ArQule has started dosing in a registrational clinical trial of derazantinib (ARQ 087) to treat patients with intrahepatic Cholangiocarcinoma (iCCA).
Derazantinib is a multi-kinase inhibitor being developed to selectively target the fibroblast growth factor receptor (FGFR).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The open-label, single-arm trial will evaluate oral, once-daily 300mg dose of derazantinib in approximately 100 subjects with FGFR2 fusion driven second-line iCCA.
Set to initially enrol US patients, the trial will later extend to European countries and will include interim analysis of data from first 40 patients who will be assessed for response.
During the trial, FGFR2 fusion status will be monitored through a break apart FISH assay, and objective response rate (ORR) will be measured as the primary endpoint.
ArQule Research and Development head and chief medical officer Dr Brian Schwartz said: “The durable response rate of 21%, we observed in the Phase I/II iCCA FGFR2 fusion driven trial has lead us to design the first registrational trial with a biomarker for this patient population.”
The registrational trial is part of a Phase II programme of derazantinib in patients with intrahepatic cholangiocarcinoma (iCCA) and FGFR2 fusions.
A Phase I portion of the programme is reported to have demonstrated two confirmed responses.
The FGFR inhibitor drug is also being studied in Phase Ib expansion cohorts for the treatment of patients with various oncology indications such as cholangiocarcinoma and adrenocortical tumours, or FGFR translocations, amplifications, and mutations.





