Global clinical-stage biotechnology firm Ascentage Pharma has formed a strategic clinical collaboration with AstraZeneca’s hematology research and development centre Acerta Pharma.
Under the partnership terms, Ascentage will sponsor a clinical trial to assess the combination of Ascentage Pharma’s APG-2575 and Acerta’s CALQUENCE (acalabrutinib).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
APG-2575 is a selective Bcl-2 inhibitor, while acalabrutinib is a Bruton’s Tyrosine Kinase (BTK) inhibitor.
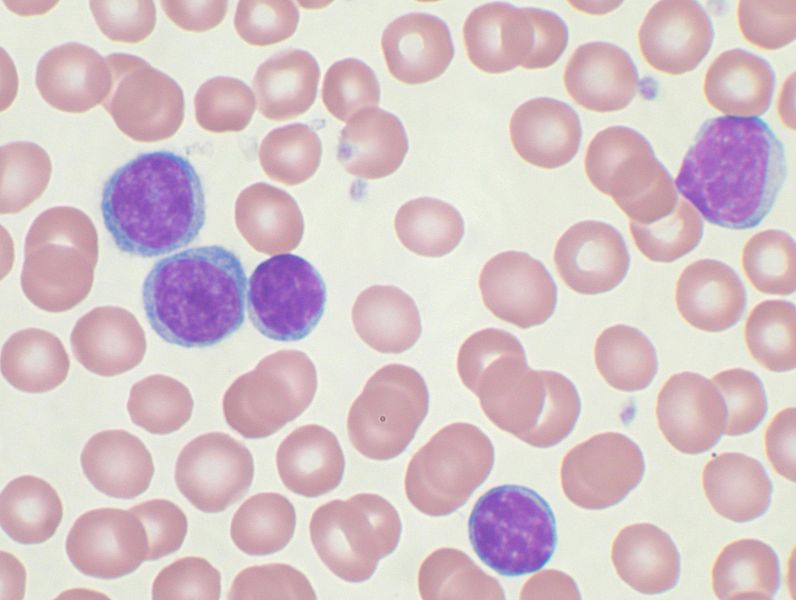
The two firms will evaluate the safety and efficacy of this combination therapy in patients identified with relapsed/refractory chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL).
According to Ascentage, the trial is a global, multicentered, open-label Phase Ib/II dose-escalation and dose-expansion study.
It is designed to evaluate the safety, efficiency, and anticancer activity of APG-2575 as a single agent or in combination with acalabrutinib in patients with relapsed/refractory CLL/SLL.
The study has already dosed its first patient in the US. It is planned to expand in Europe and Australia.
Ascentage Pharma chairman and CEO Dr Dajun Yang said: “We are delighted to enter this collaboration with Acerta. APG-2575 is a key drug candidate in our development pipeline targeting apoptosis, with great potential in the treatment of hematologic malignancies.
“Collaborating with Acerta helps to accelerate our global clinical development programme for APG-2575. Combination therapy is becoming more important in cancer treatment, and the rationale of a Bcl-2 inhibitor combined with a BTK inhibitor is sound.
“We hope that APG-2575 combined with CALQUENCE will show synergistic effects in the treatment of CLL/SLL, offering additional treatment options for patients with otherwise limited treatment options around the world.”
In July last year, Ascentage Pharma began dosing patients in a Phase I clinical trial of its Bcl-2 selective inhibitor APG-2575 to treat hematologic malignancies in China.





