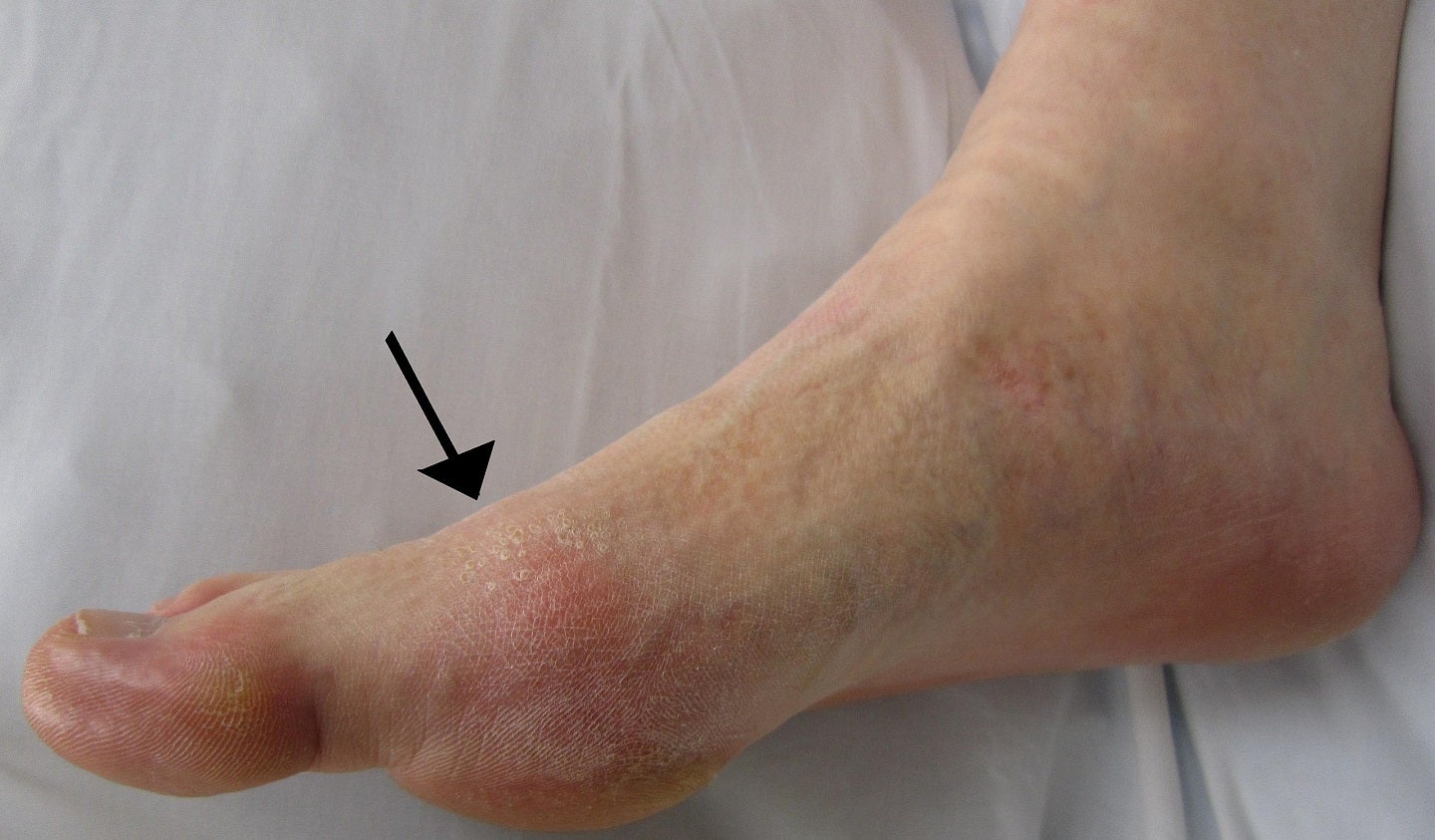
Atom Bioscience has reported positive data from its China Phase IIa study of ABP-671 for the treatment of chronic gout.
The double-blind, randomised, placebo-controlled Phase IIa clinical trial enrolled 54 patients with gout or hyperuricemia across six cohorts.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
In this trial, seven of the patients were randomised to receive ABP-671 and two to be given a placebo in each cohort.
The trial successfully met its main endpoints.
At ABP-671 1mg dose given once daily (QD), 86% of the subjects reached serum uric acid (sUA) levels that were below 6mg/dL.
Additionally, 100% of them achieved levels of sUA below 6mg/dL in several higher tested doses.
When ABP-671 QD doses of 6mg and 12mg were given, all patients reported a drop in sUA levels below 5mg/dL, and 57% and 100%, respectively, achieved sUA levels of below 4mg/dL.
All doses were found to be well-tolerated, without any significant safety concerns observed.
Atom Bioscience CEO and chairman Dr William Shi said: “The results from our Phase IIa clinical trial in China are consistent with the findings from our Phase IIa trial in Australia, indicating that ABP-671 can effectively reduce uric acid levels significantly and safely compared to currently marketed gout drugs, which have been associated with serious adverse side effects.
“The consistency of these positive results from our back-to-back clinical trials is encouraging, and we look forward to advancing ABP-671 into pivotal trials in the US and other countries later this year.”
ABP-671 is a small molecule inhibitor of the urate transporter 1 (URAT1) protein that is involved in uric acid reabsorption by the kidneys.





