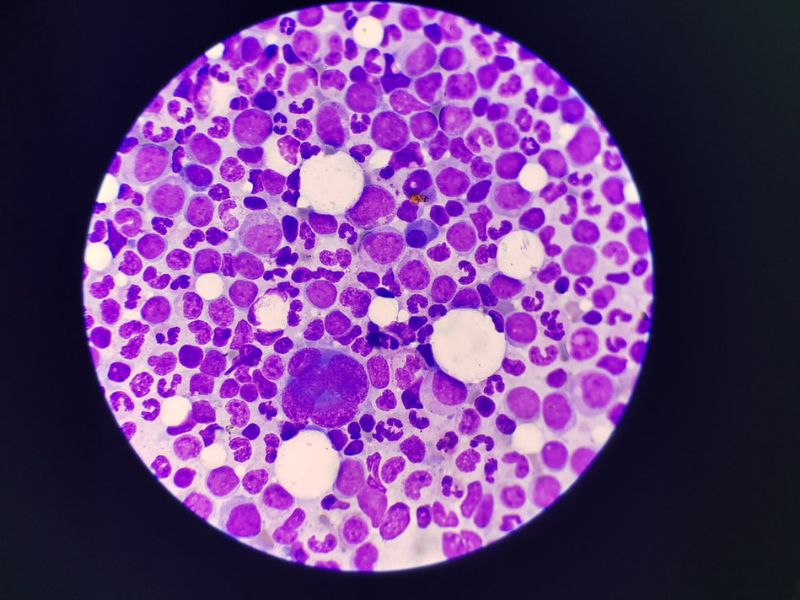
BioTheryX has initiated patient dosing in a Phase I study of BTX-A51 for the treatment of relapsed/refractory acute myeloid leukaemia (AML).
The clinical trial will assess the safety, pharmacokinetics and tolerability of BTX-A51 in patients with relapsed/refractory AML. The study will also evaluate the candidate’s efficacy on high-risk myelodysplastic syndrome patients.
BTX-A51 is a small molecule, oral multi-kinase inhibitor developed to block a specific leukemic stem cell target (CK1-alpha) and super-enhancer targets (CDK7/CDK9). The process arrests transcription of key oncogenic genes.
The inhibitor has demonstrated its potentiality for treating multiple malignancies.
In May last year, the US Food and Drug Administration (FDA) approved BioTheryX’s investigational new drug application (IND) for BTX-A51.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataBioTheryX CEO David Stirling said: “As I stated when the investigational new drug application for BTX-A51 was accepted by the FDA, the novel mechanism of BTX-A51 may become one of the most important new treatments for AML in the last 40 years, and has the potential to significantly improve the lives of AML patients and their families.”
BioTheryX said that the company was founded by a team associated with the development of the IMiDs franchise of compounds for treating cancer.
The company is also working on the development of protein degradation technology with potential applicability across a broad range of disease targets.
It utilises the body’s own protein disposal system to remove disease-causing proteins.
BioTheryX’s assets include a large number of such small molecule, orally available, cereblon-binding targeted protein degraders, referred to as protein homeostatic modulators.








