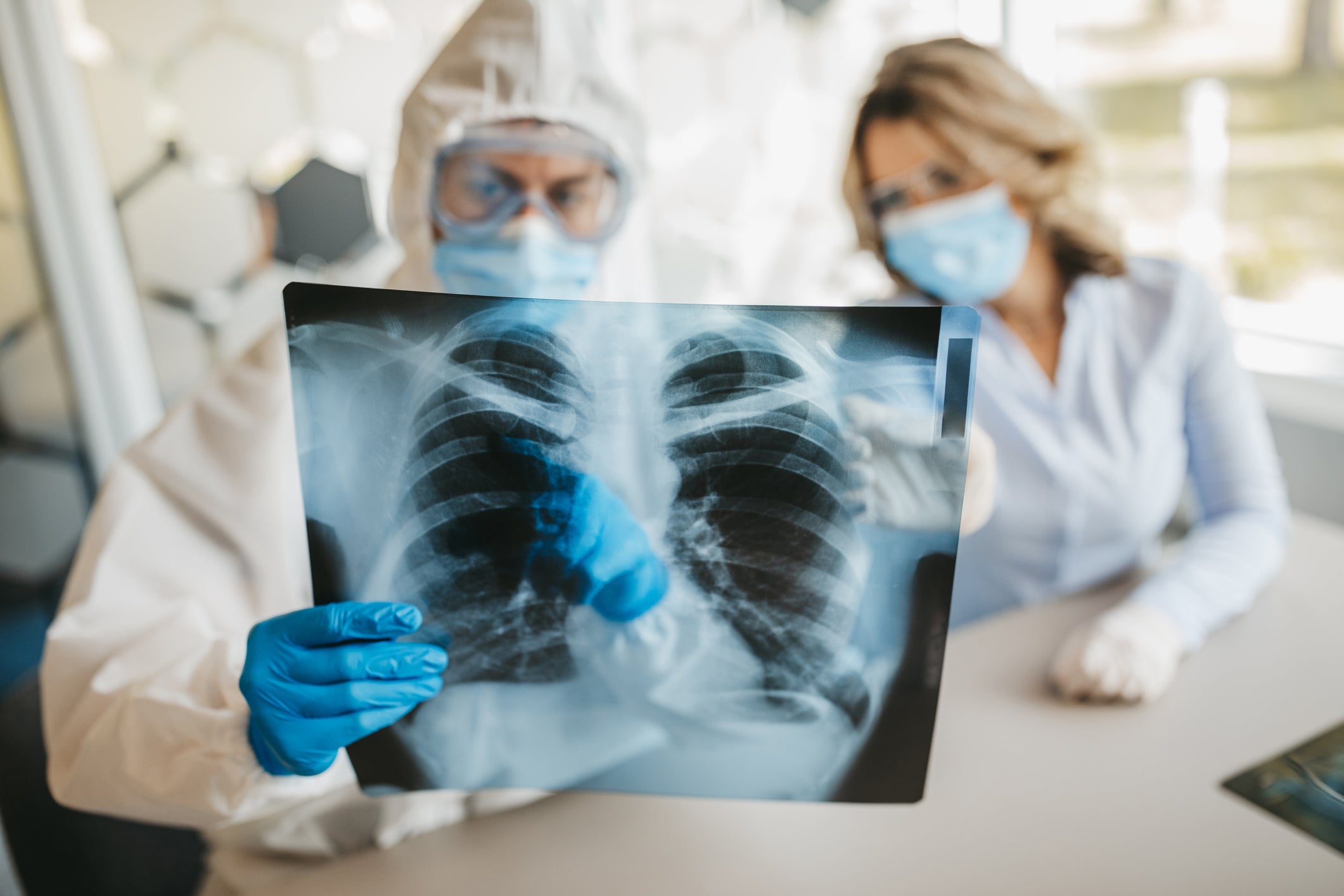
BrainStorm Cell Therapeutics has announced that its NurOwn (MSC-NTF cell) derived exosomes provided significant improvement in lung function and histology in an acute respiratory distress syndrome (ARDS) mouse model, in a preclinical study.
Mesenchymal stem cell (MSC)-derived exosomes can penetrate deep into tissues and deliver immunomodulatory molecules effectively.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
A type of respiratory failure, ARDS is linked to Covid-19 and is mediated by dysregulated cytokine production.
Intratracheal administration of NurOwn derived exosomes provided a statistically significant reduction in lung disease severity score, the study data showed.
Furthermore, improvements in lipopolysaccharide (LPS)-induced ARDS markers like lung function, fibrin presence, neutrophil accumulation, cytokine expression and oxygenation levels in the blood, were observed.
These improvements were significantly superior to those noticed following naïve MSC-derived exosome administration.
BrainStorm Research and Development vice-president Dr Revital Aricha said: “These exciting preclinical data suggest that NurOwn derived exosomes have the potential to treat Covid-19-induced ARDS or other severe respiratory complications and that they are more effective than exosomes isolated from naïve MSCs at combatting the various symptoms of the syndrome.
“This publication in a highly regarded journal provides important validation for the scientific advances and significance of BrainStorm’s preclinical research programs, including on our exosome-based technology platform.”
The NurOwn technology platform (autologous MSC-NTF cells) represents a promising investigational therapeutic approach to targeting disease pathways important in neurodegenerative disorders.
MSC-NTF cells are made from autologous, bone marrow-derived MSCs expanded and separated ex vivo.
Brainstorm CEO Chaim Lebovits said: “While our primary focus is on advancing NurOwn towards regulatory approval in ALS, we continue to evaluate the potential of our exosome-based platform to address unmet medical needs.”
In December 2019, the company received a recommendation from the independent Data Safety Monitoring Board (DSMB) to continue the Phase II clinical trial of NurOwn in progressive multiple sclerosis patients.





