Researchers from Brown University in Providence, US, have discovered a new means of stimulating autophagy, the process by which cells recycle their damaged or worn-out parts and which could provide a basis for future neurodegenerative treatments.
The research team made the discovery while examining the biological process of aging. They found autophagy to increase the lifespans of worms and flies, while experiments conducted in human cells demonstrated its potential in future treatments for neurodegenerative conditions such as Alzheimer’s disease and amyotrophic lateral sclerosis (ALS).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
“Autophagy dysfunction is present across a range of age-related diseases including neurodegeneration,” study leader Louis Lapierre said.
“We and others think that by learning how to influence this process pharmacologically, we might be able to affect the progression of these diseases. What we’ve shown here is a new and conserved entry point for stimulating autophagy,” he said.
The process of autophagy consists of misfolded proteins and obsolete organelles being collected into vesicles known as autophagosomes. These are then fused with an enzyme-containing organelle called lysosome, which breaks down the cellular macromolecules and converts them into components the cell can re-use.
The team investigated whether they could improve the autophagy process by manipulating a protein that turns gene expression on and off – known as a transcription factor – which regulates autophagic activity. As the transcription factor needs to be localised in the cell nucleus to activate autophagic activity, Lapierre’s team screened for genes that increase the autophagy transcription factor level (TFEB) within nuclei.
Using the nematode C. elegans, the team found that reducing the protein XPO1, which is used to transport proteins out of the nucleus, causes a nuclear accumulation of the nematoda version of TFEB. They found this accumulation to be linked to an increase in autophagy markers, including increased autophagosome, autolysosomes as well as increased lysosome biogenesis. The lifespan of treated nematodes were also shown to increase by between 15% and 45%.
“By blocking the escape of this transcription factor from the nucleus, we could not only influence autophagy but we could get an increase in lifespan as well,” Lapierre said.
The team found selective inhibitors of nuclear export (SINE), which are typically used in cancer treatments as XPO1-inhibitors, to have a similar gene inhibiting effect as that seen in the screening experiment.
In further experiments, SINE was tested on genetically modified fruit flies that acted as ALS models. In these experiments, the treated flies showed a small but notable increase in lifespan.
“Our data suggests that these compounds can alleviate some of the neurodegeneration in these flies,” Lapierre said.
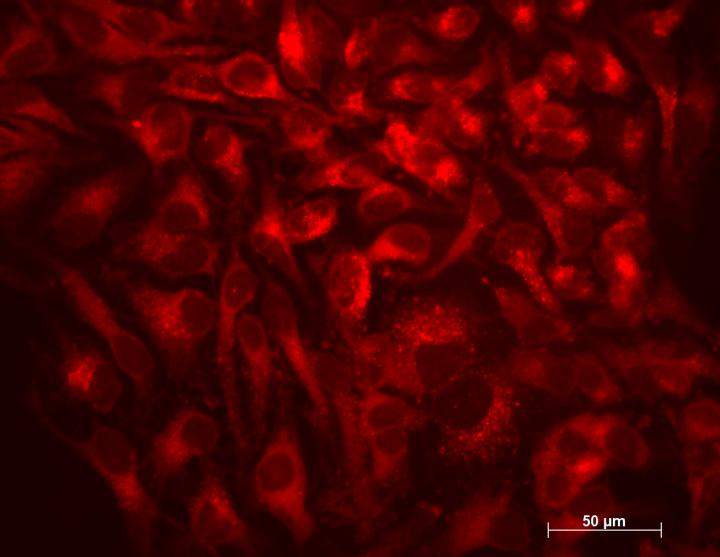
Finally, the team examined whether XPO1 inhibition had a similar effect on autophagy in human cells as in nematodes by treating human HeLa cells with SINE. Findings showed TFEB concentrations in nuclei to increase, as did markers of autophagic activity and lysosomal biogenesis.
“Our study tells us that the regulation of the intracellular partitioning of TFEB is conserved from nematodes to humans and that SINE could stimulate autophagy in humans,” Lapierre said.
“SINE have been recently shown in clinical trials for cancer to be tolerated, so the potential for using SINE to treat other age-related diseases is there.”
The initial research provides proof of concept for using autophagy as a treatment for age-related conditions. Future research is anticipated to examine these drugs in more clinically relevant models of neurodegenerative diseases.
The findings were published in the journal Cell Reports.





