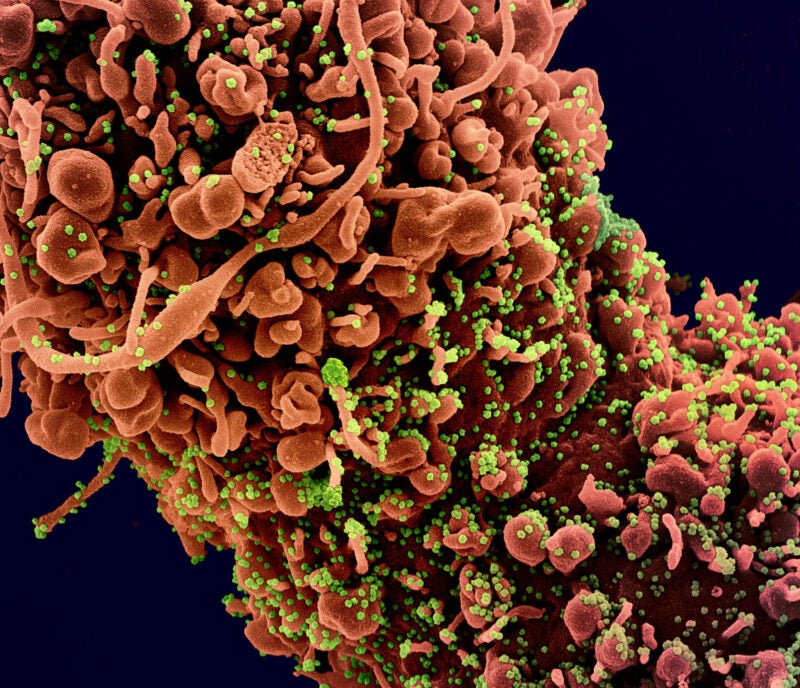
Codagenix and the Serum Institute of India (SII) have commenced subject dosing in a Phase III clinical trial of the intranasal Covid-19 vaccine CoviLiv.
The Phase III efficacy assessment is part of the Solidarity Trial Vaccines of the World Health Organization (WHO) to support second-generation Covid-19 vaccine development.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The global, multicentre, randomised, placebo-controlled trial will assess the efficacy, safety, and immunogenicity of CoviLiv against SARS-CoV-2 viral strains versus a placebo.
It will enrol up to 20,000 healthy adult subjects in countries with reduced inoculation rates on the continents of Africa, as well as in South America and Asia.
The efficacy of the vaccine against confirmed Covid-19 is the trial’s primary endpoint.
According to initial clinical findings, CoviLiv had a 100% seroresponse rate and elicited mucosal immunity that could avert nasal replication.
The vaccine was also demonstrated to induce wide-ranging cellular immune responses spanning several proteins of SARS-CoV-2, which are mostly conserved across viral variants, including those seen in Omicron BA.2.
A live-attenuated vaccine against Covid-19, CoviLiv expresses all SARS-CoV-2 proteins.
Codagenix co-founder and CEO J Robert Coleman said: “In the face of a continually evolving Covid-19 landscape, it is imperative that we bring to market vaccines that induce broad immunity, block viral shedding via mucosal immunity, and offer protection against current and future variants of SARS-CoV-2 to countries around the world.
“Data from ongoing clinical evaluations indicate that CoviLiv shows promise for meeting these important criteria, and we are thrilled WHO chose CoviLiv for this pivotal Phase III clinical evaluation.”
Apart from this Phase III trial, the companies are analysing CoviLiv as an intranasal booster in a Phase I trial in the UK.
This UK trial will assess the booster response in healthy adult subjects who were inoculated with Covid-19 vaccines earlier.
In June this year, Codagenix completed subject dosing in a Phase I trial of CodaVax-H1N1, a universal, live-attenuated influenza vaccine.





