Israel’s Rambam Medical Health Care Campus and Ziv Medical Center researchers have begun a Phase II clinical trial of Constant Therapeutics’ lead drug candidate, TXA27, for Covid-19.
A pharmaceutical formulation of the naturally occurring human peptide angiotensin-(1–7), TXA127 has an established safety profile.
The double-blind, placebo-controlled trial will enrol 120 subjects who are hospitalised with moderate Covid-19 and who do not require invasive ventilation. A total of 21 participants have so far been enrolled in the trial.
Half of the participants will receive the TXA127 treatment, while the other half will be given a placebo.
Meanwhile, the US Food and Drug Administration (FDA) has granted an IND for a second Phase II trial being carried out at Columbia University Irving Medical Center and New York-Presbyterian Allen Hospital.
This double-blind, placebo-controlled trial will have 100 subjects.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataThe company noted that 3% of Covid-19 patients in the US and Israel get hospitalised and many do not respond to some standard therapies, such as steroids, antivirals and monoclonal antibodies.
Constant Therapeutics CEO Dr Richard Franklin said: “In addition to researchers in Israel and at Columbia, we have been contacted by medical centres and scientists in the US, UK, Belgium, Italy, Spain and Australia suggesting the use of TXA127 for the treatment of Covid-19 and requesting drug for trials.”
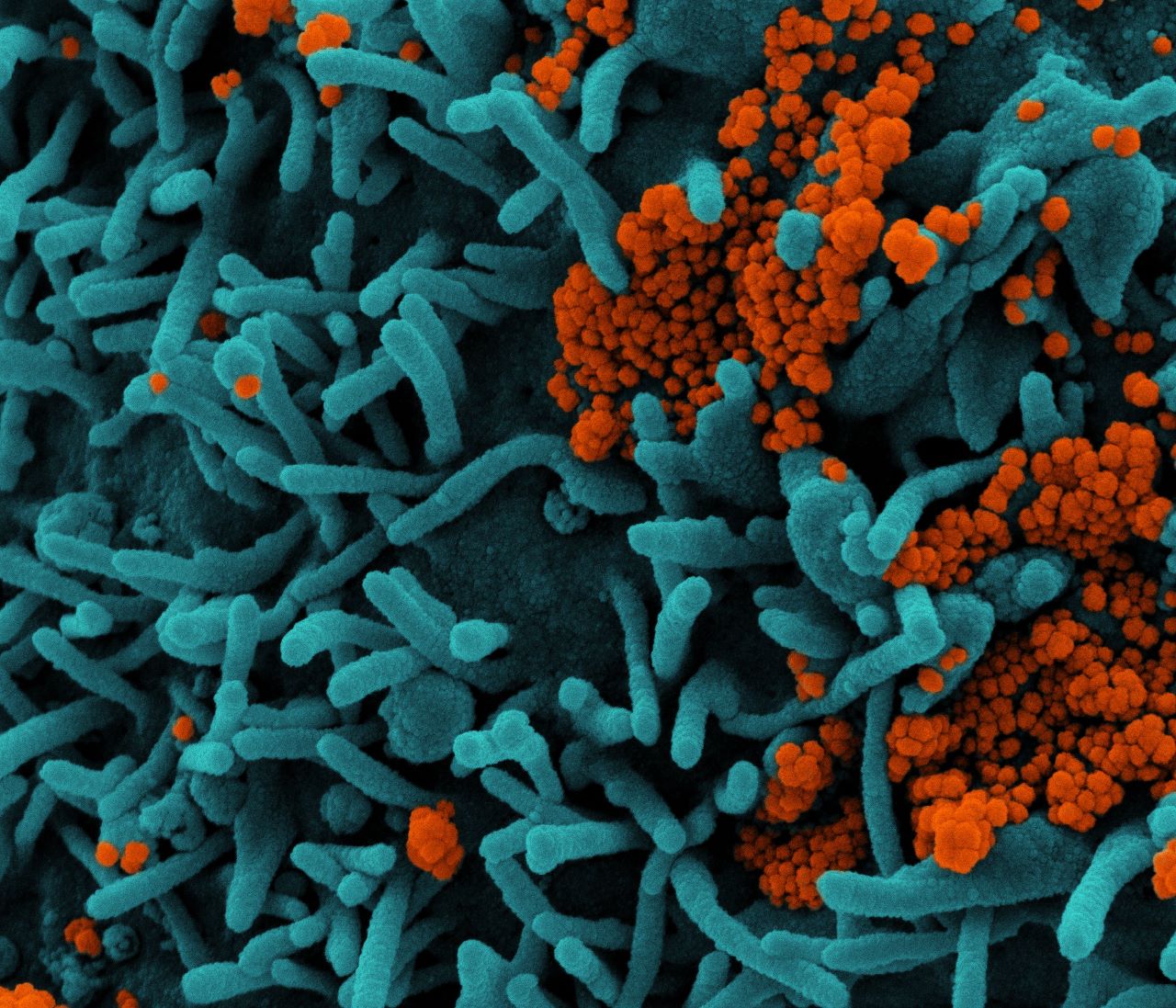
“All of the current variants, including those appearing in the UK, South Africa, and Brazil, although behaving differently in terms of transmissibility and susceptibility to vaccines, still attach to ACE2 and should be treatable by TXA127.”
The trial in Israel will be completed in four months.
The company is currently evaluating the potential use of TXA127 in treating more severely ill patients in the ICU and newly infected people in the outpatient setting using an oral formulation of the drug candidate.







