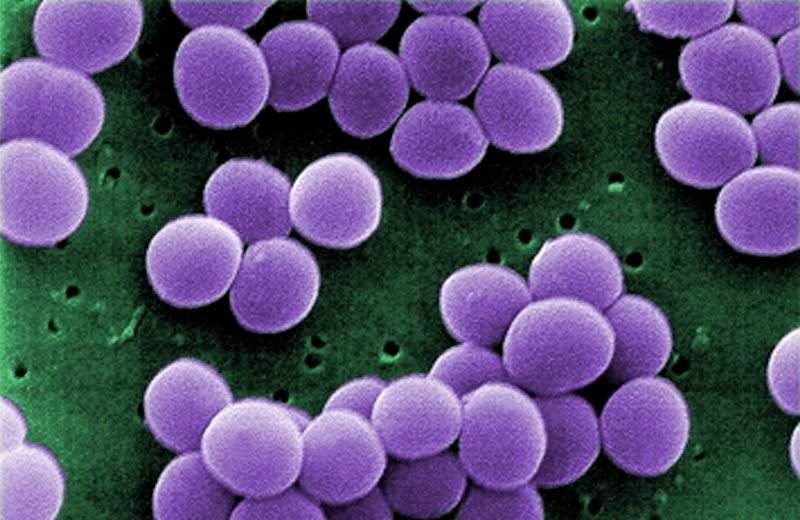
ContraFect has completed enrolment in the Phase ll clinical trial examining CF-301 (exebacase) as a potential treatment for Staphylococcus aureus (Staph aureus) bacteremia, including endocarditis, a serious infection that affects the heart valves.
The multi-centre, multi-national, randomised, double-blind, placebo-controlled superiority trial is designed to investigate CF-301, along with standard-of-care (SOC) antibiotics and in comparison with SOC alone, to improve clinical cure rates in the enrolled patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The trial is currently being conducted at various sites in the US, Europe, Latin America, Russia and Israel.
As part of the trial, around 115 patients are expected to be randomised in a 3:2 ratio to receive either a single dose of CF-301 or placebo administered via intravenous infusion in addition to SOC.
The trial’s primary endpoints are to analyse the safety, tolerability, pharmacokinetics, and efficacy of CF-301.
ContraFect CEO and Research and Development executive vice-president Cara Cassino said: “Staph aureus bacteremia and endocarditis continue to be associated with substantial morbidity and mortality despite conventional antibiotics.
“We look forward to the topline results of the Phase ll study later this year, which will inform us of the potential for CF-301 to improve clinical outcomes for these serious, potentially life-threatening Staph aureus infections.”
CF-301is a recombinant bacteriophage-derived lysin with effective bactericidal activity against Staph aureus, which causes bloodstream infections, or bacteraemia.
It features a new, rapid, and specific mechanism that can take bactericidal action against Staph aureus without impacting the body’s natural bacterial flora.
According to ContraFect, CF-301 plus standard of care antibiotics has already proved to have significantly increased bacterial killing and survival in animal models of disease when compared to antibiotics or CF-301 alone.





