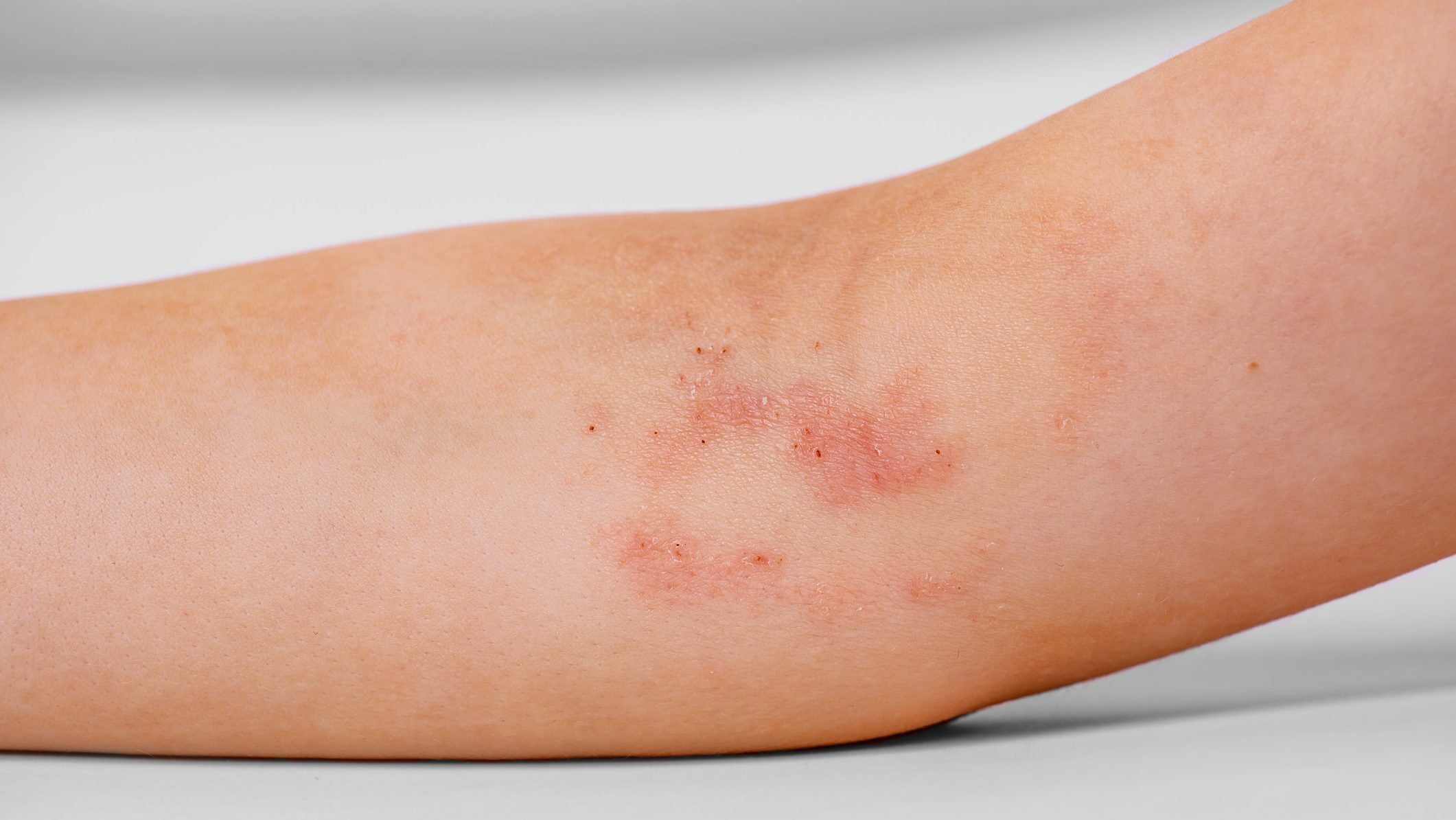
US-based pharmaceutical company Dermavant Sciences, a division of Roivant Sciences, has announced positive results from its Phase III development programme evaluating Vtama (tapinarof) cream for the treatment of atopic dermatitis.
The development programme is made up of three studies that are evaluating the 1% Vtama cream in adults and paediatric patients over the age of two.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
ADORING 1 and ADORING 2 are double-blind, randomised trials that assessed the safety and efficacy of the cream, while ADORING 3 is an open-label, long-term extension study that will evaluate the cream for up to 48 weeks.
Dermavant conducted an integrated analysis, which combines data from all three studies and a Maximal Usage Pharmacokinetics (MUPK) study.
The integrated analysis revealed that out of the 711 patients, 80.7% achieved at least a 75% reduction in disease burden. Within 24 hours of the first application, 77.9% of patients aged 12 and over experienced itch reduction. Also, an interim analysis of the ADORING 3 study demonstrated a 51.2% complete disease clearance rate.
In the announcement accompanying the data, Dermavant’s CEO Todd Zavodnick explained the company’s plans to submit an sNDA: “We are focused on compiling our sNDA data for submission that is expected in Q1 of 2024 and we look forward to making VTAMA cream, 1% available as a potential treatment option for both adults and children suffering from atopic dermatitis as expeditiously as possible, subject to regulatory approval.”
Vtama is an aryl hydrocarbon receptor agonist (AHRA), which is administered as a once-daily, steroid-free topical cream.
In 2022, Dermavant gained US Food and Drug Administration approval for the treatment of plaque psoriasis in adults after submitting a new drug application in May 2021.
Vtama generated $28m for the fiscal year ending 31 March 2023, as per Roivant’s Q4 and year-end 2023 financial results.
The North Carolina-headquartered company gained the rights to Vtama from GSK back in 2018 in a $250m deal. Under the agreement, Dermavant gained all global rights to the cream, with the exception of China.





