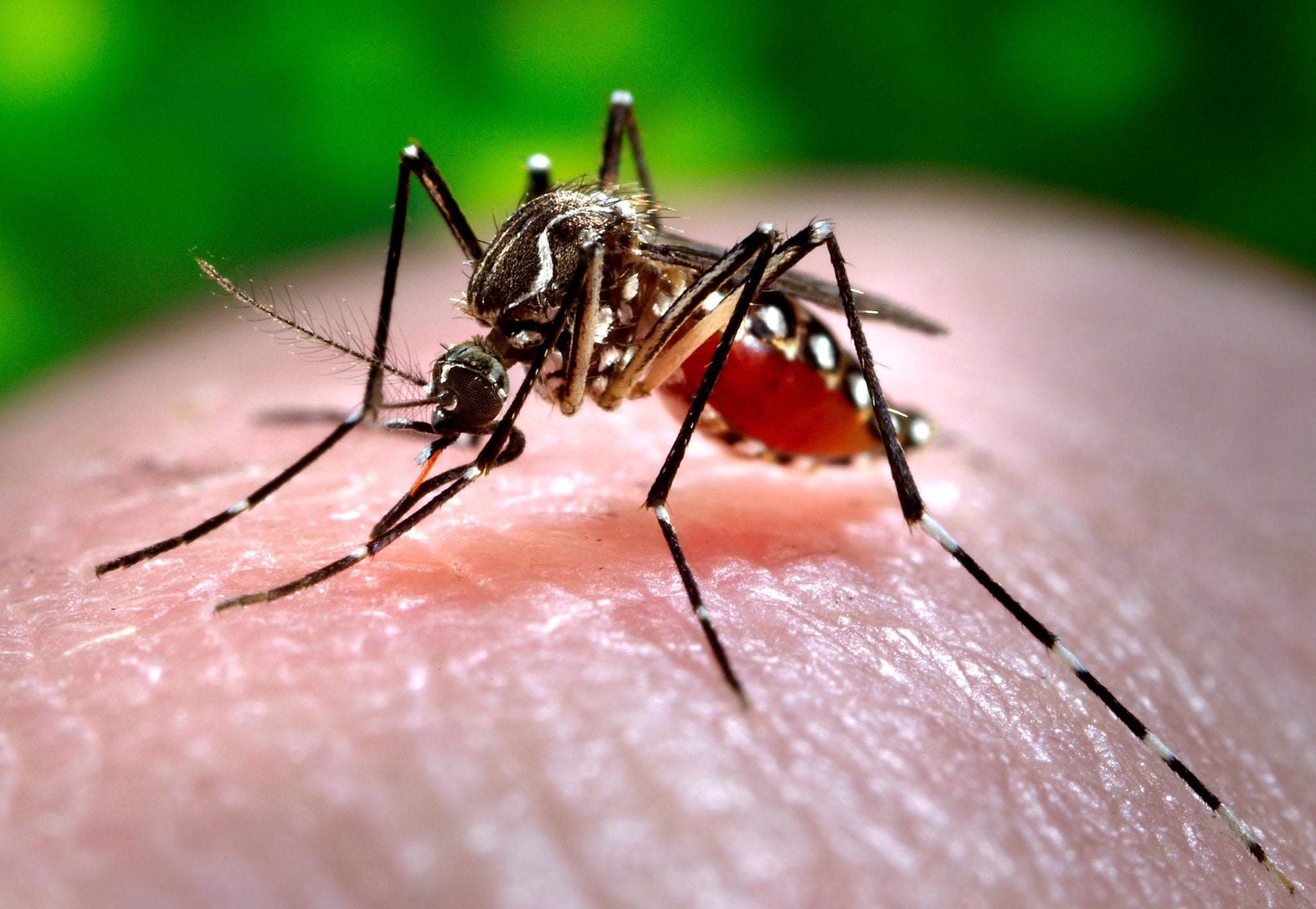
Emergent BioSolutions has dosed the first subject in the Phase III clinical trial of its single dose investigational chikungunya virus (CHIKV) vaccine candidate, CHIKV VLP.
It is being developed for active immunisation against chikungunya disease, which is spread to humans by virus infected mosquitoes. CHIKV VLP is claimed to be the only virus-like particle (VLP) -based vaccine.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The multicentre, double blind, randomised, placebo-controlled Phase III trial will assess the safety and immunogenicity of the CHIKV VLP vaccine candidate in healthy people aged 12 to 64.
The trial intends to enrol a minimum of 3,150 subjects in up to 49 centres across the US.
It will also evaluate the consistency of the CHIKV serum neutralising antibody (SNA) response across three lots of manufactured vaccine.
Furthermore, the trial will analyse the CHIKV SNA response at day 22 in terms of geometric mean titer and seroresponse rate.
The US Food and Drug Administration granted breakthrough therapy status and fast track status to the vaccine in October last year and May 2018, respectively.
In September 2019, CHIKV VLP also obtained PRIME status from the European Medicines Agency.
Emergent BioSolutions executive vice-president and chief medical officer Karen Smith said: “Emergent has achieved a major milestone as we begin our Phase III study for our single-dose chikungunya vaccine candidate.
“I am proud of the Emergent team for bringing us a step closer to potentially having a critical solution to address this important disease for which no vaccine or treatment is currently available.”
Emergent reported two-year persistence data early this year from its Phase II trial of the vaccine in terms of safety and immunogenicity in 415 healthy adults.
CHIKV VLP was found to have a favourable safety profile.
Most of the adverse events were mild or moderately severe, with local injection site pain being the most frequent one.
Furthermore, two years after vaccination with adjuvanted 40µg dose of CHIKV VLP, SNA responses were found to be 19-fold greater than pre-vaccination titers. This data backs the durability of the immune response, the company said.
All participants in the single-dose regimen were found to be seropositive during their one-year and two-year visits.
In April last year, Emergent reported positive data from the interim analysis of a Phase II trial of the CHIKV-VLP vaccine candidate.





