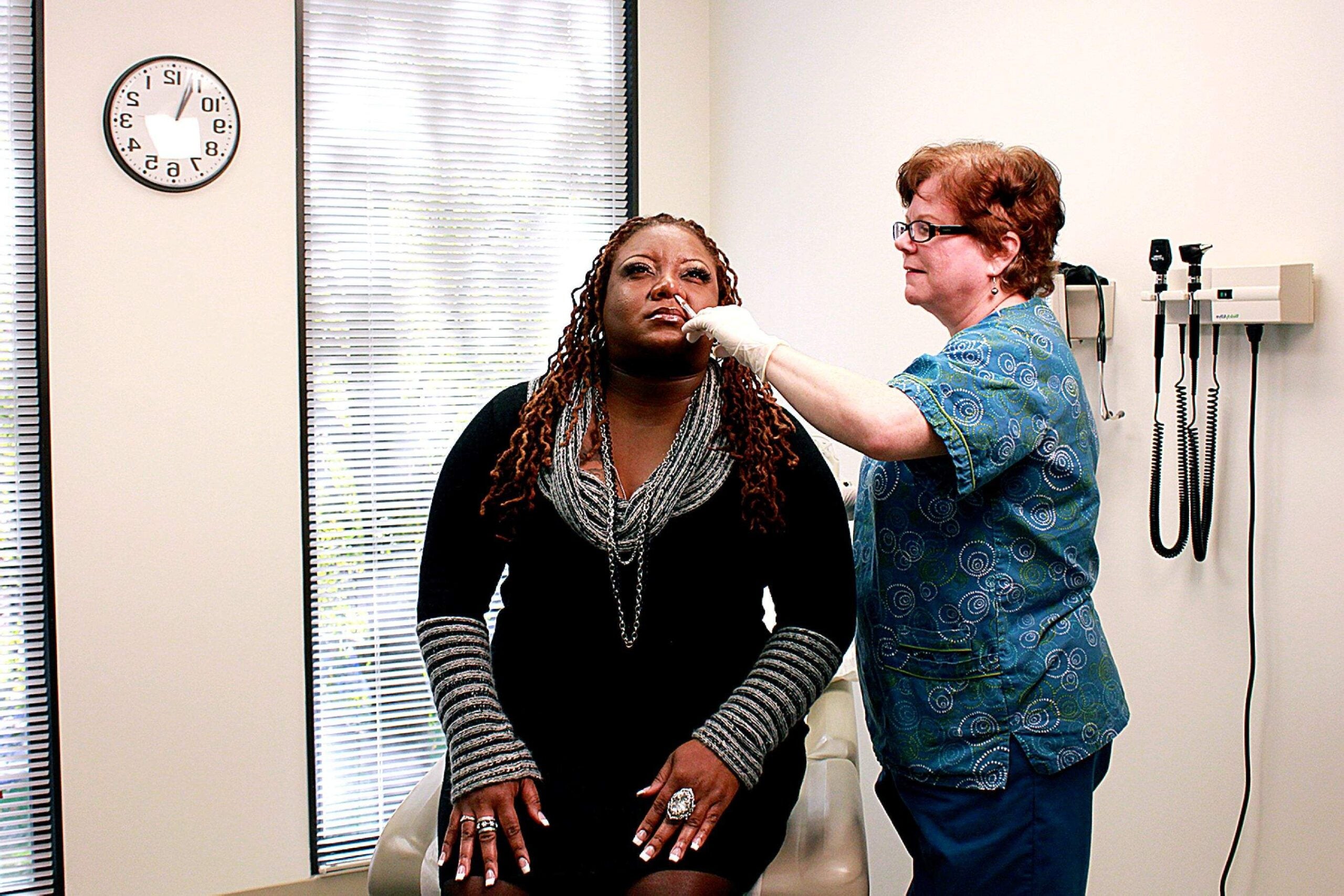
Evoke Pharma has announced that the European Patent Office has agreed to patent its treatment Gimoti, targeting diabetic gastroparesis specifically seen in women.
The patent grant applies to 38 covered member states and is due to be published on 9 May this year, with patent expiry extended to 2032.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
“This patent represents the first female-specific intellectual property approved for Gimoti and highlights the significant work we’ve accomplished in developing this novel new treatment for gastroparesis,” Evoke CEO Dave Gonyer said.
“Based on our research findings, we have also filed other formulation, efficacy, pharmacokinetic, and dosing patents directed toward the specific profile in women in order to further solidify our intellectual property position.”
Diabetic gastroparesis is a gastrointestinal (GI) disorder in which the stomach takes too long to empty its contents as a result of erratic or slow muscular contractions. This causes severe digestive system symptoms such as nausea, vomiting, abdominal pain and bloating.
Gastroparesis often hinders oral medications’ ability to pass through the stomach and be properly absorbed by the body. Additionally, vomiting as a result of the condition can impair the efficacy of oral medications. In extreme cases patients can experience malnutrition and require hospitalisation. If left untreated, it can also lead to acute medical problems such as increased diabetic complications which result from poor glucose control.
It is estimated that around 16 million people suffer from gastroparesis globally, with 80% of this population being female.
Evoke is known for its work developing treatments for GI diseases. Gimoti is the company’s nasal formulation of metoclopramide, a drug for the relief of symptoms associated with acute and recurrent gastroparesis in women with diabetes mellitus.
Metoclopramide is the only treatment for the condition currently approved in the US. Approximately four million prescriptions of oral metoclopramide are given each year in the US, and as such a non-oral formulation would provide a much needed alternative for patients. The nasal spray is designed to bypass the stomach and enable the effective delivery of the medication directly into the bloodstream.
Evoke has announced its plans to submit a New Drug Application (NDA) for Gimoti to the US Food and Drug Administration (FDA) within the second quarter of 2018.





