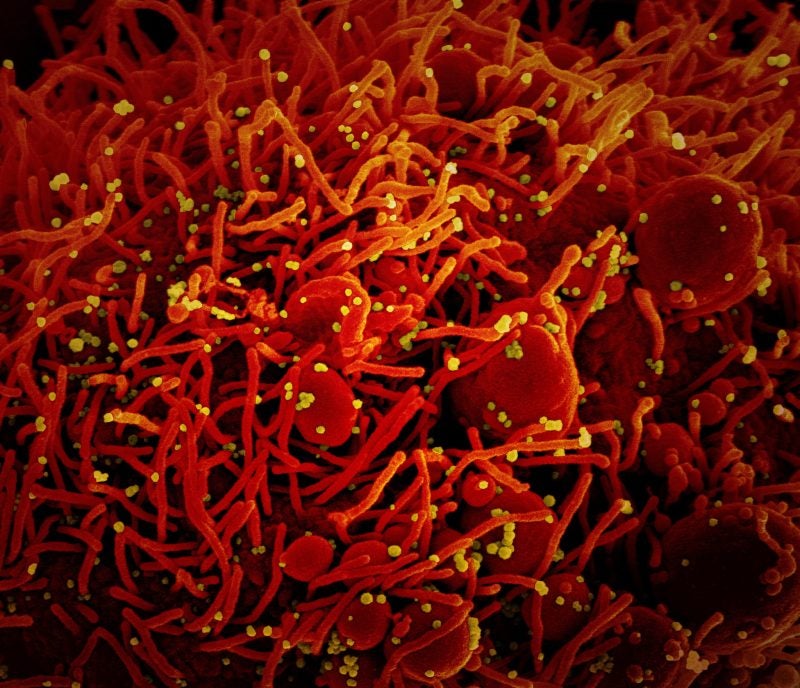

The US Food and Drug Administration (FDA) has granted clearance to Sorrento Therapeutics to commence clinical trials of the messenger ribonucleic acid (mRNA) vaccine STI-1557.
Targeting the Omicron variants of the SARS-CoV-2 virus, the vaccine incorporates a mutation in the furin cleavage site that prevents the S1 subunit cleavage from the spike protein from attaching to the muscle cell membrane.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
This variation was introduced to prevent S1 subunit leakage from the expressed spike protein (S protein) into the bloodstream.
S1 subunit leakage is claimed to possibly cause unwanted side effects in tissues of vital organs, following dosing with mRNA vaccines approved by the FDA at present.
For protecting mRNAs from degradation, they are encased in a Sorrento lipid nanoparticle formulation (LNP) and facilitate the S protein’s translation in host cells, following intramuscular (IM) dosing, thereby eliciting an adaptive immune response.
Using the Sofusa MuVaxx lymphatic delivery system, the company intends to seek more sophisticated mRNA vaccine approaches in the future.
The microneedle system facilitates the intradermal delivery of the vaccines.
Along with an mRNA vaccine candidate, this system showed enhanced humoral immunity and increased cellular immunity using one tenth of the IM dose in animal models.
According to the data from the preclinical research using MuVaxx, immune responses were induced and lasted at a ten-fold reduction in dosage versus standard IM dosing, as evaluated by anti-spike antibodies, cytokine-producing CD8 T cells, and neutralising antibodies.
In addition, following MuVaxx administered inoculation, a four-fold rise in T cell response was seen versus IM delivery.
In August this year, Sorrento announced plans to begin a Phase I clinical trial of its oral therapy, STI-1558, for Covid-19 in China.
The move comes after the company obtained the China National Medical Products Administration (NMPA) approval for its IND application for STI-1558.
Cell & Gene Therapy coverage on Clinical Trials Arena is supported by Cytiva.
Editorial content is independently produced and follows the highest standards of journalistic integrity. Topic sponsors are not involved in the creation of editorial content.

