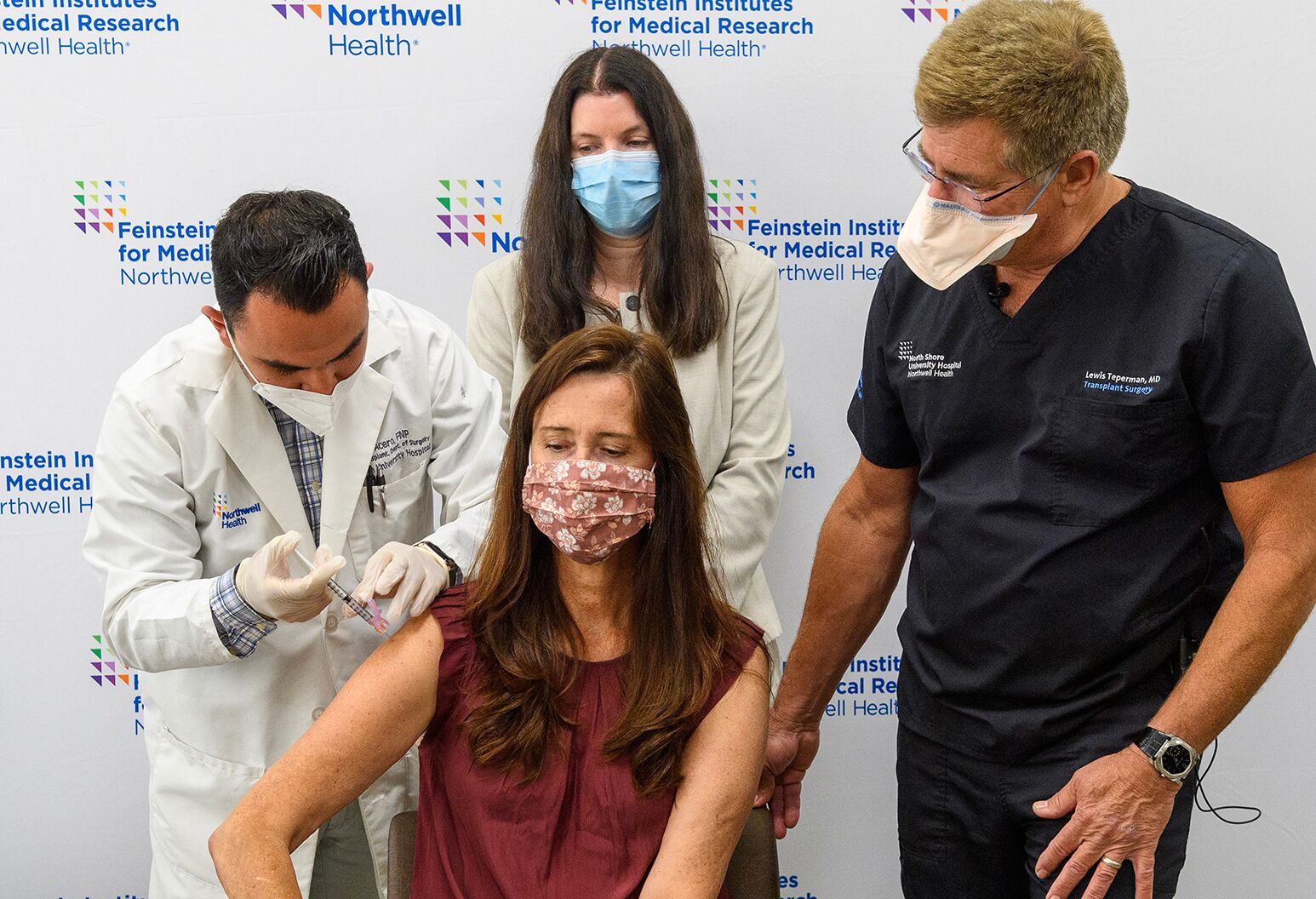
Northwell Health’s science arm Feinstein Institutes for Medical Research in the US has commenced the clinical trial of a third dose of Moderna’s Covid-19 vaccine in individuals living with an organ transplant.
Sponsored by Moderna, the multicentre, nationwide trial will assess the efficacy of a third shot administered following the initial two-dose regimen in patients in the country.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Individuals who have had an organ transplant are at an increased risk of contracting Covid-19 and developing severe disease.
Immunosuppressive treatments used after an organ transplant could hinder the recipient’s ability to develop a robust immune defence against SARS-CoV-2 by administering a two-dose vaccine regimen, Feinstein noted.
The trial intends to enrol 240 subjects, including healthy participants, with the Feinstein Institutes being one of the seven trial centres leading the study.
It will analyse the immune antibody response of the transplant patients 28 days from the third dose.
Feinstein Institutes clinical research vice-president Christina Brennan said: “Since the start of the Covid-19 pandemic, the Feinstein Institutes focused on conducting clinical trials in an effort to find safe and effective treatments.
“Now, with the support of Moderna, we hope this vaccine clinical trial will give insight into the best way to protect a high-risk population.”
In March 2020, the Feinstein Institutes launched its initial set of trials to analyse the safety and efficacy of potential Covid-19 treatments.
The institute commenced subject enrolment in another trial in the US to test the antibody response on receiving booster shots of authorised or approved vaccines in patients with an autoimmune disease.





