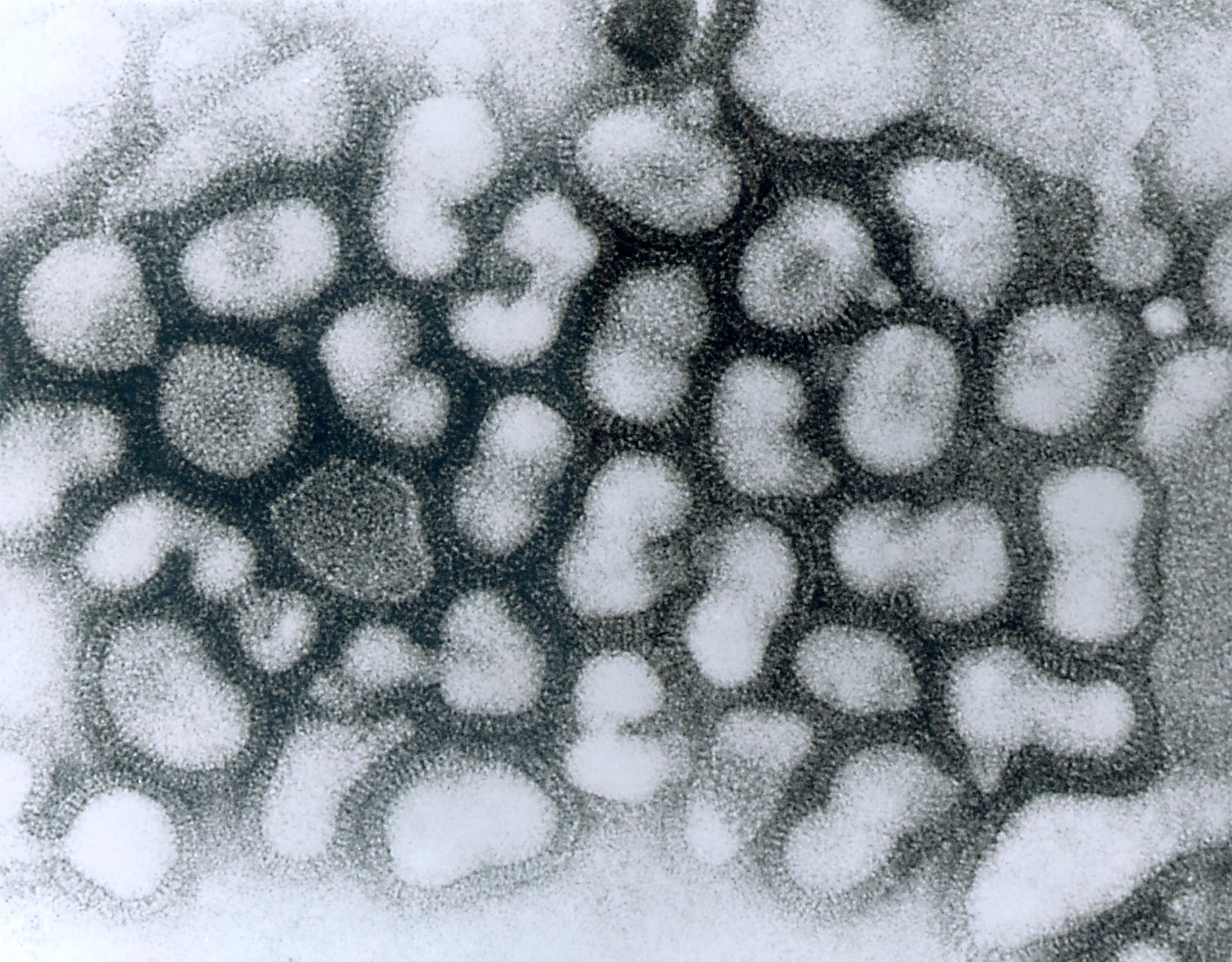
FluGen has begun the Phase Ib study of its investigational intranasal influenza (flu) vaccine, M2SR, in healthy adults aged 50 to 85.
A lead candidate of FluGen, the supra-seasonal, live, single-replication vaccine can potentially induce mucosal, humoral and cellular immunity.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Named FluGen H3N2 v005, the randomised, double-blind, placebo-controlled Phase Ib trial will examine the safety, tolerability and immunological response to a monovalent Singapore 2016 H3N2 M2SR influenza A vaccine.
FluGen noted that the participating age group is prone to develop serious complications and mortality from contracting flu.
The study, funded by the US National Institutes of Health (NIH) unit National Institute of Allergy and Infectious Diseases, will have 120 participants in three successive age cohorts of 50-64, 65-85 lead-in, and 65-85 expansion.
Participants will be given a single intranasal dose of M2SR or placebo followed by a single dose of inactivated quadrivalent influenza vaccine (QIV) after 28 days in all cohorts.
The safety and tolerability of the vaccine after six months of inoculation will form the trial’s primary goal while the safety profile of QIV given after M2SR will be one of the secondary endpoints.
Furthermore, the trial will analyse the immunogenicity as determined by serum and mucosal antibody responses of a single dose of Sing 2016 M2SR to similar and drifted flu virus strains.
FluGen president and CEO Paul Radspinner said: “We have already demonstrated the ability of M2SR, our supra-seasonal, live, single replication, intranasal flu vaccine, to protect healthy adults when challenged with a seven-year drifted influenza strain.
“Now, in this highly vulnerable population, we aim to further show immunogenicity against matched and mismatched virus strains, as well as a broad immune response, including mucosal immunity, which is not typically seen with mRNA or inactivated vaccines.”
The company anticipates reporting top-line data from the trial in the first half of next year.





