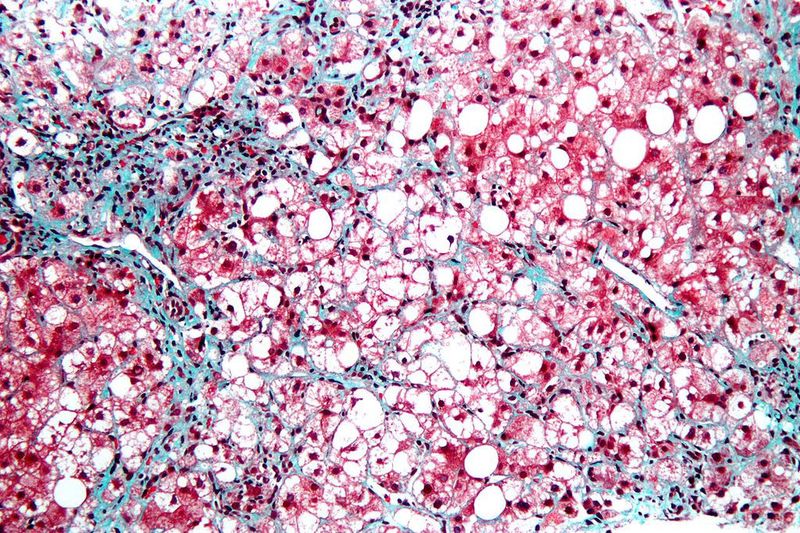
Top-line data reported by Gilead Sciences has revealed that the Phase II ATLAS clinical trial of cilofexor, firsocostat and selonsertib did not meet the primary endpoint in nonalcoholic steatohepatitis (NASH) patients with bridging fibrosis (F3) and compensated cirrhosis (F4).
The 48-week randomised, double-blind, placebo-controlled trial assessed the drugs as monotherapy and dual combination regimens in 392 patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Each of the drugs exhibits different mechanisms of action. Cilofexor is a nonsteroidal farnesoid X receptor (FXR) agonist, firsocostat is an acetyl-CoA carboxylase (ACC) inhibitor, and selonsertib is an apoptosis signal-regulating kinase 1 (ASK1) inhibitor.
According to the results, no regimen demonstrated a statistically significant increase in the proportion of participants with a ≥1-stage improvement in fibrosis without worsening of NASH, which was the primary efficacy endpoint of the study.
However, the firsocostat and cilofexor combination led to statistically significant improvements in various fibrosis and liver function measures compared to placebo.
In the trial, the selonsertib monotherapy group was discontinued after the termination of the STELLAR trials.
The drugs were generally well-tolerated as monotherapies and dual combination regimens in the ATLAS study. The most common adverse events with firsocostat plus cilofexor were mild to moderate pruritus, headache, diarrhoea and nausea.
Gilead Sciences chief medical officer Merdad Parsey said: “NASH is a complex disease driven by multiple mechanisms. The results from the ATLAS study suggest the potential for a combination therapeutic approach for patients with advanced fibrosis by targeting different aspects of this disease.
“We continue to analyse the ATLAS data and will work with regulators to determine appropriate next steps for these therapies.”
Gilead is also evaluating cilofexor and firsocostat in combination with Novo Nordisk’s GLP-1 analogue semaglutide to treat NASH patients.





