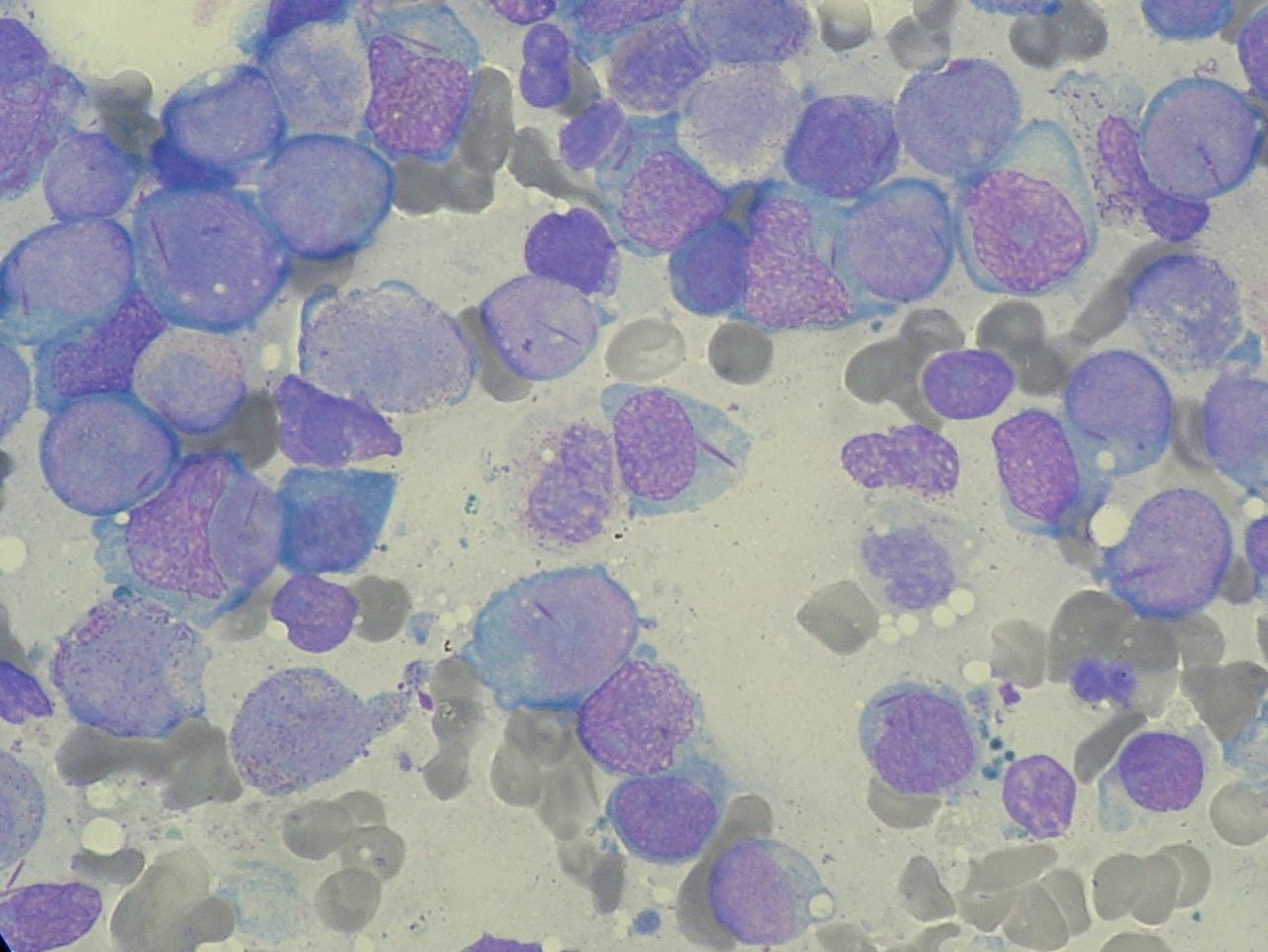
Taiwanese biopharma company Golden Biotechnology (GoldenBiotech) has reported that data from its Phase IIa clinical study conducted in Russia showed its new drug Antroquinonol (Hocena) outperformed the other listing drugs for treating relapsed acute myeloid leukaemia (AML) patients.
The outcome measures showed higher remission rates and survival rates, assuring fewer patients will need blood transfusions.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The open-label, non-randomised, Phase IIa trial analysed the efficacy and safety/tolerability profiles of Antroquinonol as monotherapy in relapsed AML patients or at initial diagnosis when no intensive treatment is possible.
Patients took oral doses of 200mg twice a day at home.
According to the primary efficacy indicator results, the overall remission rate (CR/CRi) of patients was at 50% as compared to other listing drugs with CR/Cri at 21% to 33%.
Also, 80% of patients who were given the treatment showed that their abnormal blasts in bone marrow or blood were less than 5%.
About 90% of them did not need blood transfusion during the trial period and all patients were all alive during the trial period of six months.
No significant side effect (SAE) were observed during the treatment period.
GoldenBiotech plans to submit an investigational new drug (IND) application to the US Food and Drug Administration (FDA) for the Phase II clinical trials of Antroquinonol as the first-line treatment of AML.
Apart from this, Antroquinonol is being evaluated in a Phase II clinical trial for treating mild to moderate Covid-19 in hospitalised patients in the US.
This is the first Taiwanese new drug approved by FDA for the Phase II clinical trial in Covid-19.
In previous preclinical studies, it demonstrated anti-viral, anti-inflammatory and anti-fibrotic characteristics, hinting at its potential in becoming a successful treatment option for Covid-19 induced pneumonia.





