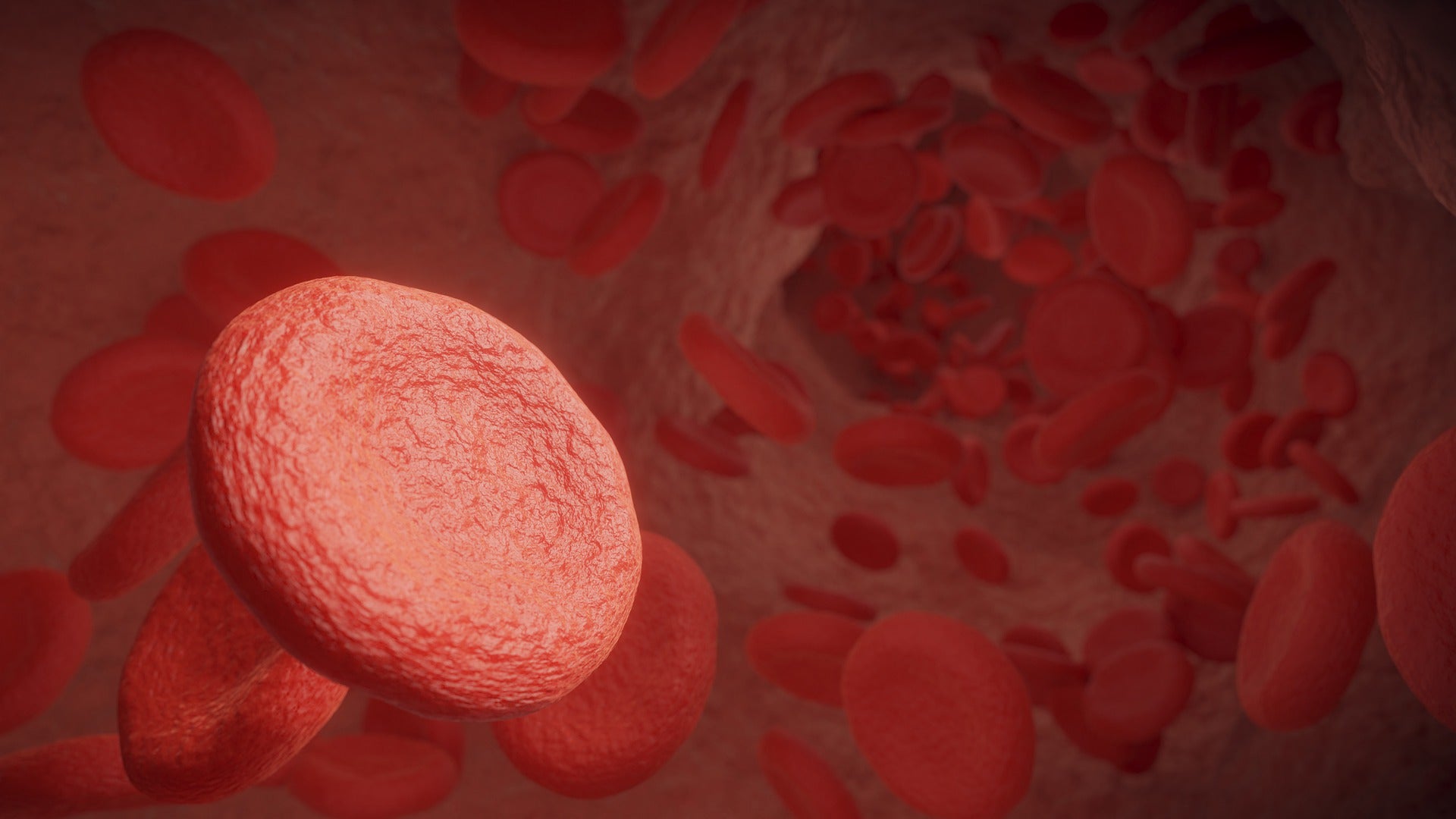
Janssen Pharmaceutical has reported follow-up results from Phase III ANDROMEDA study where its Darzalex Faspro (daratumumab and hyaluronidase-fihj) offered improved haematologic complete response (hemCR) in newly diagnosed light chain (AL) amyloidosis patients.
Darzalex Faspro is a CD38-directed antibody that received accelerated approval from the US Food and Drug Administration in combination with bortezomib (Velcade), cyclophosphamide and dexamethasone (VCd) for treating AL amyloidosis patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
This FDA approval is based on initial data, including response rate findings, from the Phase III study.
AL amyloidosis is a rare blood cell disease that affects the function and causes deterioration of vital organs, mainly the heart, kidneys and liver.
The ongoing, randomised, open-label Phase III ANDROMEDA trial is assessing the safety and efficacy of Darzalex Faspro plus VCd (D-VCd), versus VCd alone, in 388 adult patients with AL amyloidosis.
With a median follow up of 20.3 months, longer-term data demonstrated rates of hemCR were increased in subjects receiving D-VCd versus VCd alone.
At a median follow-up of 11.4 months, hemCR was observed to be 53% and 18% in D-VCd arm and VCd arm, respectively, while at 20.3 months, the rates rose to 59% and 19%, respectively.
Furthermore, 79% of subjects in the D-VCd arm showed a very good partial response or better compared to 50% in the VCd arm after 20.3 months of median follow-up.
No new safety signals of Darzalex Faspro were noted in the trial while the most common adverse reactions were found to be diarrhoea, peripheral oedema, constipation and peripheral sensory neuropathy, among others.
Janssen Research & Development Clinical Development vice-president Jessica Vermeulen said: “These updated efficacy and safety results show the potential impact Darzalex Faspro can have in treating AL amyloidosis.
“Following the FDA approval earlier this year, these latest data are a result of our commitment to further exploring the potential of Darzalex in haematologic and rare diseases for patients in need of new options.”
This development comes after Janssen reported follow-up results from the Phase I MajesTEC-1 dose-escalation study, where its investigational antibody, teclistamab, provided durable responses in relapsed or refractory multiple myeloma patients.





