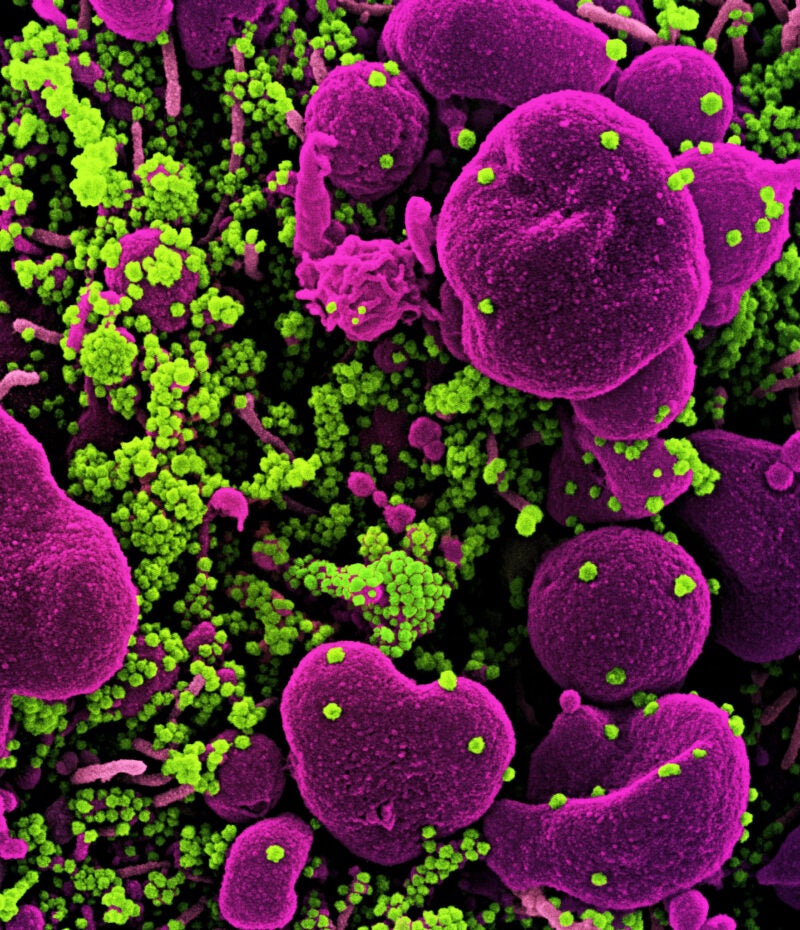
Kinarus Therapeutics has announced plans to discontinue the Phase II KINETIC clinical trial of KIN001 in Covid-19 patients in the hospital setting.
The latest development comes after the independent Data and Safety Monitoring Board (DSMB) carried out a review of the study data and recommended the trial discontinuation.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
With a less than expected occurrence of the primary endpoint due to the evolution of the existing therapy landscape, the board decided that there exists a reduced chance to demonstrate a statistically significant benefit with an acceptable number of Covid-19 patients in the hospital.
As a result, the DSMB recommended the discontinuation of the trial.
According to the safety results from the trial, a favourable profile was reported, with the occurrence of treatment-emergent adverse events (TEAEs) balanced between the trial arms.
A combination of pamapimod and pioglitazone, KIN001 showed synergistic efficacy and enhanced response durability in preclinical models of several disease indications.
In cells infected with SARS-CoV-2, hindering p38 using the combination therapy showed to lower the replication of the virus, indicating synergistic activity compared to single therapies.
KIN001 could also dull the overactive inflammatory response in ambulatory Covid-19 patients to lower disease duration, hospital admission, long-term morbidity, and the occurrence of long Covid.
Kinarus Therapeutics CEO Dr Alexander Bausch said: “The underlying scientific rationale for KIN001 remains intact as p38 MAP kinase, its key biological target, is well characterised and implicated in other indications via biological mechanisms that lead to a variety of chronic inflammatory diseases, including wet age-related macular degeneration (wAMD) and idiopathic pulmonary fibrosis (IPF).
“The DSMB recommendation has no impact on our plans to develop KIN001 for treatment of wAMD and IPF or other indications.”
In August this year, the company dosed the first subject in the Phase II KINFAST trial of KIN001 in mild or moderate Covid-19 patients in an outpatient setting.





