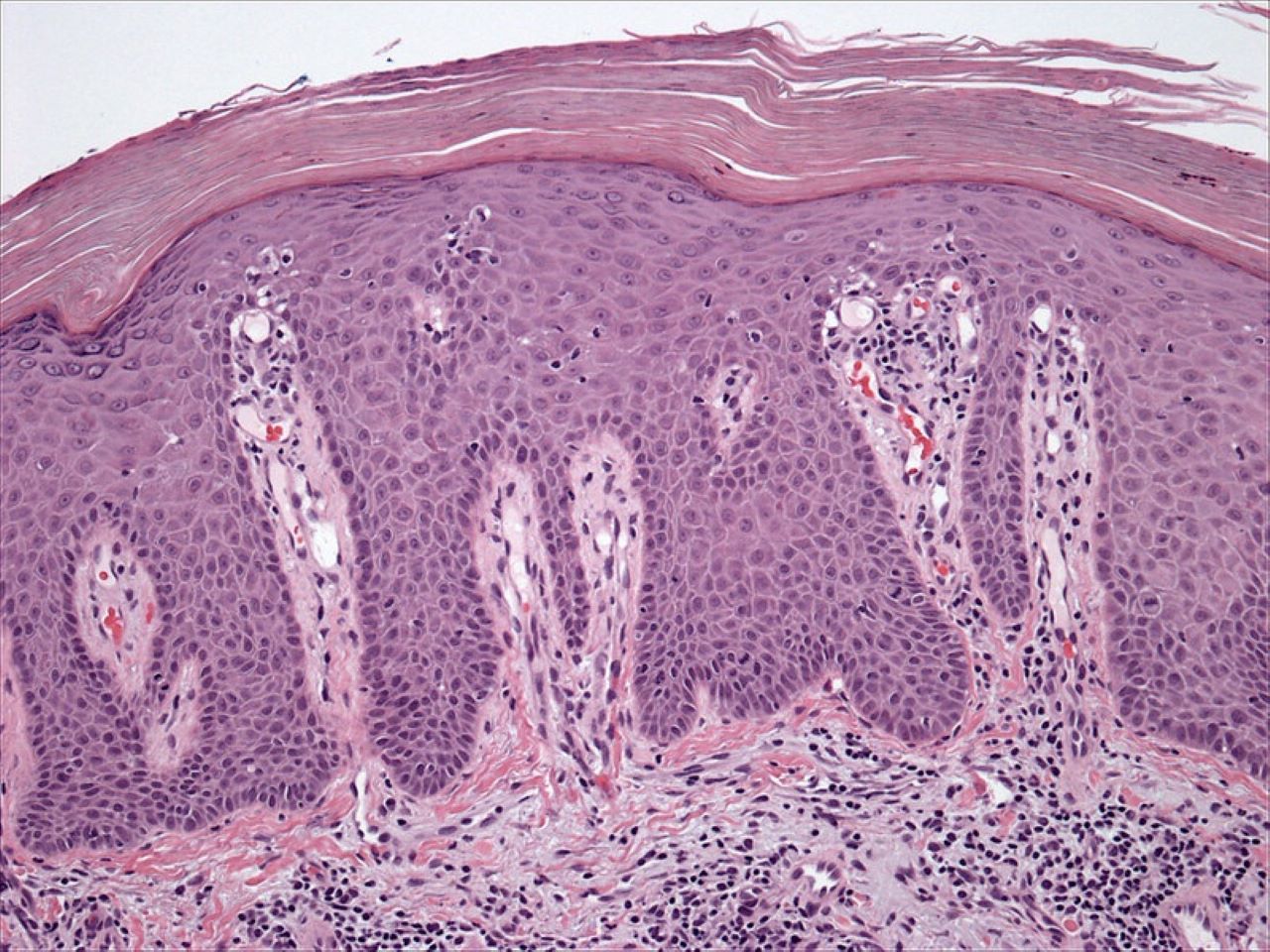
Eli Lilly and Company has reported that multiple long-term and post-hoc analyses of the monoclonal antibody Taltz (ixekizumab) showed sustained efficacy in patients with psoriasis and psoriatic arthritis.
This includes results from the extension period of UNCOVER-3 showing five-year sustained efficacy of Taltz in patients with psoriasis.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
In the UNCOVER-3 trial, adult patients were randomised across four treatment arms and given placebo or Taltz.
They initially received Taltz 80mg every two weeks or every four weeks following an initial dose of Taltz 160mg or etanercept 50mg twice weekly in the 12-week induction period.
At week 12, eligible patients entered the long-term extension period and were given the US FDA-approved dose of 80mg Taltz every four weeks.
Results from the trial showed that 83%, 73% and 89% of patients reported complete clearance in scalp, nail and palmoplantar psoriasis, respectively, at week 264.
They also reported mean per cent improvements of 89%, 88% and 95% from baseline in scalp, nail and palmoplantar psoriasis, respectively.
The safety profile of the drug over five years was consistent with previously reported ones with no new adverse events.
In the SPIRIT-P2 trial, Taltz demonstrated persistent clearance of nail and skin psoriasis plaques for up to three years in tumour necrosis factor inhibitors (TNFi) treatment-naïve patients with active psoriatic arthritis.
A post-hoc analysis was carried out on patients with nail psoriasis at baseline from the SPIRIT-H2H trial of TNFi treatment-naïve patients with both active psoriatic arthritis and active psoriasis.
It showed complete resolution of nail psoriasis was maintained for up to 52 weeks in 83% of patients treated with Taltz compared to 72% in those who received adalimumab.
Eli Lilly and Company immunology development vice-president Lotus Mallbris said: “These long-term studies provide healthcare providers and people living with psoriasis and psoriatic arthritis valuable information regarding the effectiveness of Taltz in helping patients achieve and sustain complete skin clearance, even in these particularly burdensome skin areas.”





