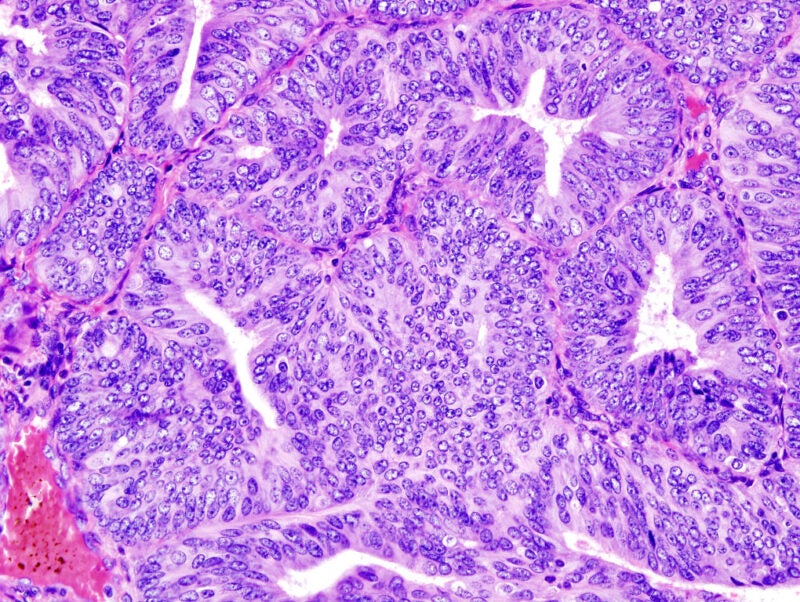
Mersana Therapeutics has commenced the dosing of subjects in the Phase I clinical trial of XMT-1660 to treat individuals with solid tumours, including breast, endometrial, and ovarian cancers.
The multicentre trial will assess XMT-1660’s safety, tolerability, and anti-tumour activity in such solid tumour patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The preliminary dose escalation part of the trial will analyse the safety and tolerability of XMT-1660 as a monotherapy.
In the dose expansion segment of the trial, the tolerability and efficacy of XMT-1660 will be evaluated.
Investigator-evaluated objective response rate and duration of response are the primary endpoints of this portion of the trial.
XMT-1660 is a Dolasynthen antibody-drug conjugate (ADC) that acts on B7-H4.
It has a target-streamlined drug-to-antibody ratio and the company’s DolaLock microtubule inhibitor payload established clinically, with a controlled bystander effect.
B7-H4 is overexpressed in various cancers, including breast, endometrial, and ovarian tumours.
The therapy also showed strong anti-tumour activity in various models representing each of these cancer types and in multiple patient-obtained xenograft models in pre-clinical studies.
Mersana Therapeutics president and CEO Anna Protopapas said: “The initiation of this Phase I trial represents an important milestone for Mersana as we continue to build our pipeline across three ADC platforms and seek to further demonstrate the potential of our Dolalock payload and Dolasynthen platform.
“Based on our promising preclinical data, we believe XMT-1660 has the potential to serve as a highly impactful cancer treatment.”
The clinical-stage biopharma firm discovers and develops ADCs that act on cancers in areas of increased unmet medical need.





