Moleculin has undertaken and enrolled patients in two anti-cancer clinical trials in Poland.
Moleculin recently recruited three patients in six weeks in Poland and has finished building a similar cohort in the US.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
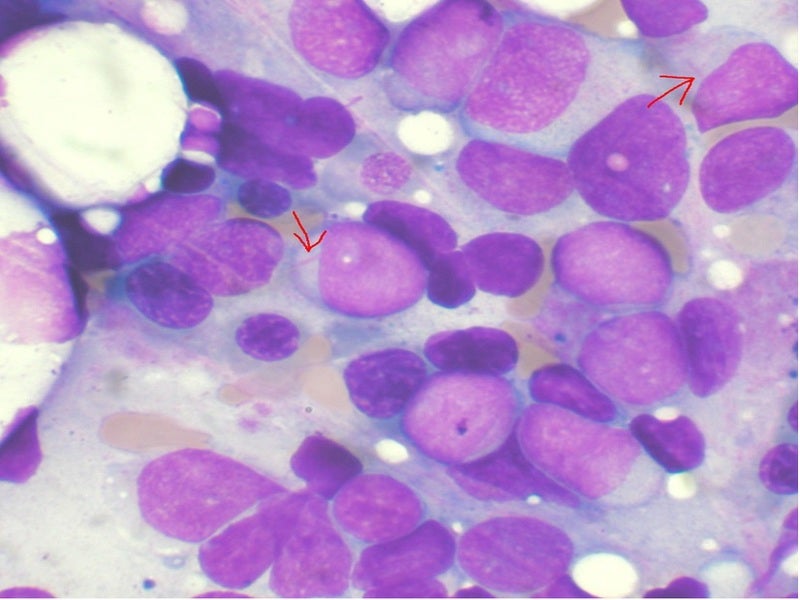
The company’s Phase I/II clinical trial is currently underway in Poland to evaluate its Annamycin compound and has already reported positive data, with one patient’s acute myeloid leukaemia (AML) now in remission.
Moleculin has already started to identify more patients in Poland for the next cohort of the same trial and is planning to enrol two more patients for its WP1220 topical treatment of cutaneous T-cell lymphoma (CTCL).
Moleculin CEO Walter Klemp said: “For a smaller pharmaceutical company with a promising roster of drug candidates for targeting these highly resistant tumours, speed in launching trials is critical.
“We have seen no evidence of cardiotoxicity in any of the patients treated thus far and intend to advance the clinical study of Annamycin with the goal of ultimately demonstrating the drug’s safety and effectiveness to support regulatory approval in the US and European Union.”
With the additional patient safety data gained from the Polish trials, FDA will be able to review cardiac safety, which can be a key factor in regulatory approval.
Moleculin started working with the medical community in Poland in order to build a larger base of clinical trial patients quickly.
The allowable starting dose for the current Moleculin Polish trial was 120mg/m², offering the opportunity to provide patients with a potentially more therapeutic dose than the 100mg/m² required in the US.
The second cohort will begin in Poland at a dose level of 150mg/m² after three patients complete the safety evaluation period of the first cohort.
Moleculin signed an agreement with Emory University earlier this month to conduct a Phase I clinical trial of WP1066 in children with recurrent or refractory malignant brain tumours.





