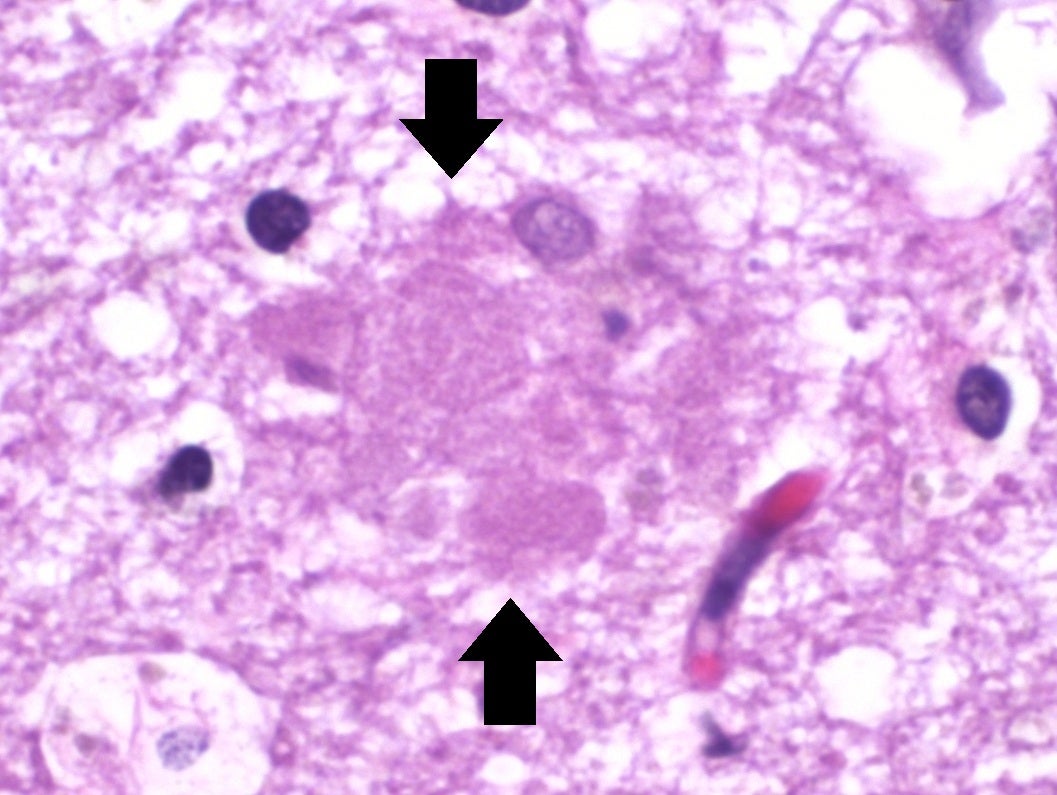
Biopharma company Neurotrope has dosed the first patient in its long-term Phase II trial of lead therapeutic candidate Bryostatin-1 to treat Alzheimer’s disease (AD).
Backed by a $2.7m grant, the trial plans to enrol around 100 patients and will be conducted in collaboration with the National Institutes of Health (NIH).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The study will analyse Bryostatin-1 in the absence of Namenda (memantine) for six months including two 11-week dosing cycles.
Participants will include AD patients with pre-specified moderately severe and moderate disease, including those who showed evidence of benefit in the previous study.
The study will also assess sustained cognitive benefit as measured by the Severe Impairment Battery (SIB) score, a measure of cognitive function in advanced dementia patients.
Neurotrope president and chief scientific officer Dr Daniel Alkon said: “After reviewing data from previous trials of Bryostatin-1 with both key opinion leaders and the NIH, we determined that a study to evaluate its long-term therapeutic effects in the absence of Namenda in patients with AD was a priority.”
This study is backed by Phase II clinical data from a pilot trial, which assessed Bryostatin-1 in the absence of Namenda for 11 weeks.
Results showed that a 20mcg dose of the drug was well tolerated and demonstrated early signals of cognitive benefit, which included improvement in SIB score by 5.0 points as compared to baseline in the Moderate Stratum cohort in the non-Namenda group.
This SIB score improvement was consistent throughout the treatment period and persisted for four weeks even after treatment ended.
A second pilot trial using the same treatment protocol showed a similar SIB improvement.





