US-based clinical-stage company bluebird bio has begun treatment in the Phase I clinical trial (HGB-206) of its LentiGlobin BB305 drug product to treat patients with severe sickle cell disease (SCD).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Designed to treat transfusion-dependent β-thalassemia and severe sickle-cell disease, LentiGlobin BB305 is a product candidate that leverages the firm's lentiviral gene delivery and T-cell engineering.
The ongoing, open-label Phase I trial will evaluate the safety and efficacy of LentiGlobin BB305 in the treatment of up to 29 subjects with SCD.
The study will assess the changes in red cell function tests, hemolysis markers, and frequency of clinical events secondary to SCD such as vaso-occlusive crises or acute chest syndrome.
bluebird bio chief medical officer David Davidson said: “This research has informed numerous changes we have implemented in the HGB-206 study protocol that, in addition to the introduction of the transduction enhancers into our manufacturing process, we hope will improve in vivo VCN and HbAT87Q expression.
"The impressive drug product VCN achieved in the first patient under this amended protocol highlights the success of the changes to our drug product manufacturing process, and we are hopeful that these modifications will improve patient outcomes.”
The firm has incorporated multiple changes to the study protocol, such as increasing the percentage of transduced cells, cell processing and myeloablation, as well as introducing a minimum period of regular blood transfusions before stem cell collection.
The study will also explore an alternate hematopoietic stem cell (HSC) procurement method. These changes are expected to enhance the production of therapeutic anti-sickling hemoglobin (HbAT87Q).
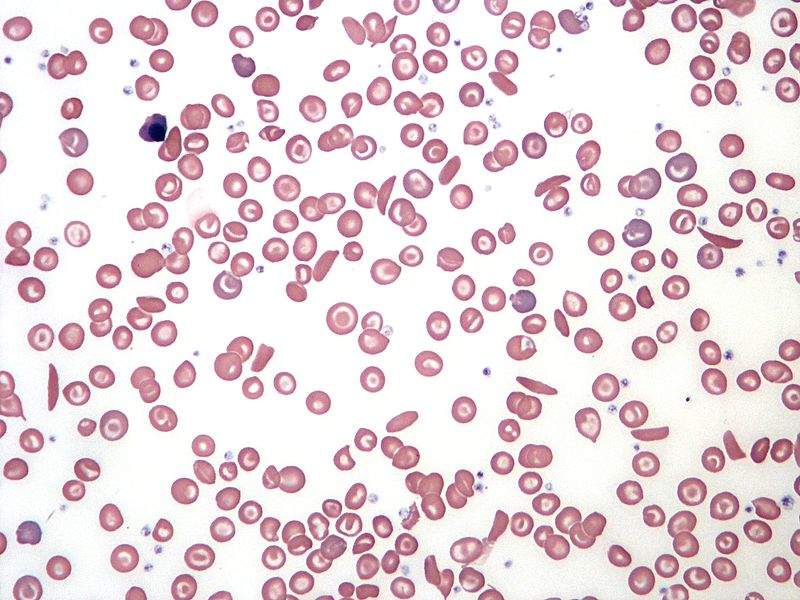
Image: Blood smear illustrating sickle cell anaemia. Photo: courtesy of Keith Chambers/Wikipedia.





