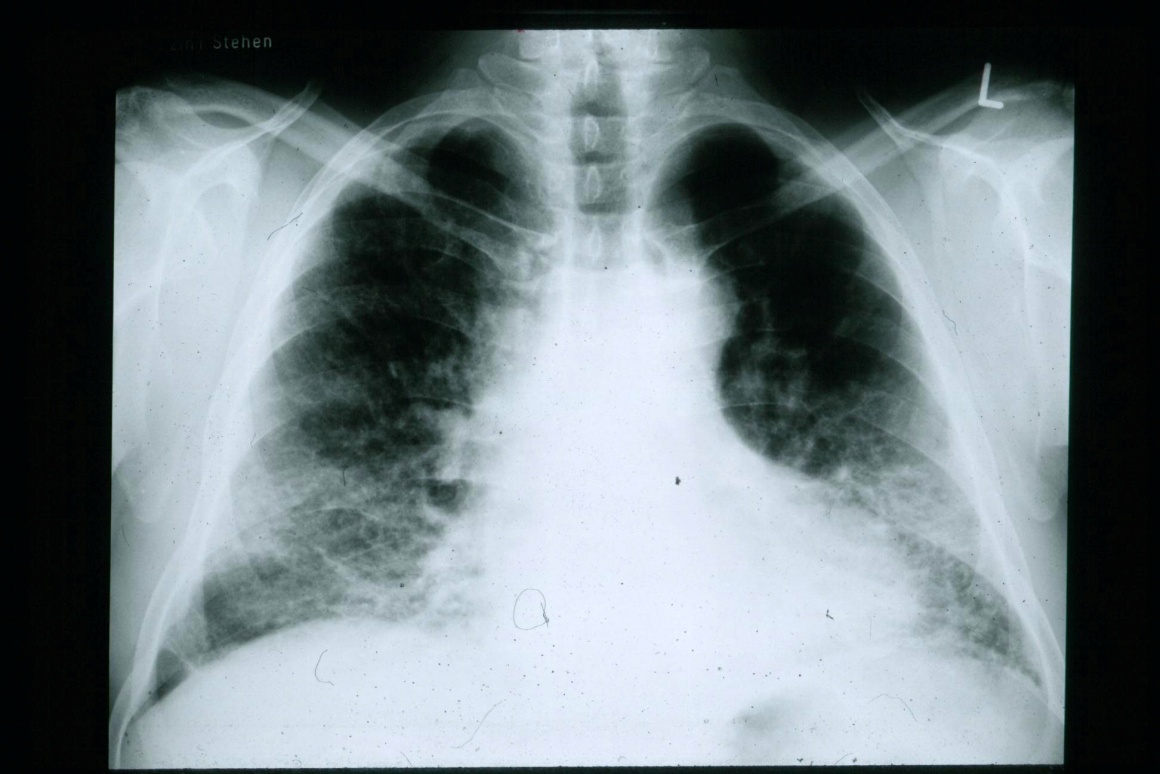
German pharmaceutical company Boehringer Ingelheim has enrolled the first patient in its new study, INMARK, to evaluate OFEV (nintedanib) on specific blood biomarkers in patients with idiopathic pulmonary fibrosis (IPF).
OFEV (nintedanib) is a small molecule tyrosine kinase inhibitor developed by Boehringer Ingelheim to treat IPF.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
INMARK has been designed to evaluate the safety and tolerability of OFEV when administered in combination with other drugs and across different patient populations.
Royal Brompton Hospital consultant respiratory physician and study principal investigator Dr Toby Maher said: "The trial is of great scientific value in that it will provide better understanding of IPF and the value of nintedanib treatment in individuals with preserved lung function.
"Importantly this is the first time we are investigating the effect of any anti-fibrotic treatment on changes in specific biomarkers in IPF.
"Being able to identify biomarkers that will predict how the disease will progress in a specific patient will allow physicians to start their patients on appropriate treatment at the earliest opportunity to help slow disease progression."
Identification of changes in biomarkers indicates the excessive scarring (fibrosis) in the lungs of patients with IPF.
Biomarkers are indicative of the presence or severity of a condition and are used to monitor and predict the disease course of patients, which will pave the way for an appropriate treatment.
INMARK is designed to determine the changes in various extracellular matrix (ECM) turnover biomarkers.
IPF is a debilitating and fatal lung disease that records a high percentage of mortality.
OFEV has been able to slow the advance of the disease, however it further necessitates the identification of biomarkers to determine the progression of the disease, as well as identify treatments suiting specific patients.
Image: Chest radiograph displaying IPF. Photo: courtesy of IPFeditor via Wikipedia.





