
US-based pharmaceutical company Corcept Therapeutics has reported positive data from its Phase I/II trial of CORT125281 by adding mifepristone to the compound to treat patients with metastatic triple-negative breast cancer (TNBC).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
CORT125281 is one of the selective cortisol modulators that the company is evaluating to treat Cushing’s syndrome.
Mifepristone is an anti-progestin inhibiting the action of progesterone, a hormone necessary to maintain a pregnancy.
It constitutes an active ingredient of Corcept’s US Food and Drug Administartion (FDA) approved product, Korlym, which is an orally administered, once-daily treatment for hyperglycemia, secondary to endogenous Cushing’s syndrome.
The Phase I/II trial was conducted to determine the efficacy of addition of mifepristone to denibulin (Halaven) in patients with TNBC whose tumours express glucocorticoid receptor (GR), one of the receptors to which mifepristone binds.
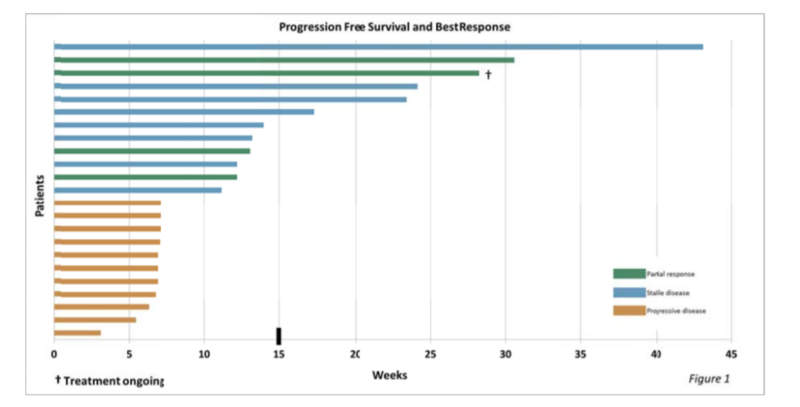
The study examined 21 patients with GR positive tumours, one with a GR negative tumour and one whose GR status is not known.
It was observed that four patients experienced partial response in the form of 30% or greater reduction in tumour size. Eight patients had stable disease and 11 had progressive disease.
The results were determined on the basis of response evaluation criteria in solid tumours (RECIST).
Corcept chief medical officer Robert Fishman said: “Six patients achieved progression-free survival (PFS) longer than the upper bound for PFS (15 weeks) in patients receiving Halaven monotherapy in a comparable population.
“Median PFS in our trial was 11.1 weeks compared to 7.2 weeks in the Halaven monotherapy study reported by Aegis. We continue to collect overall survival data. Eight study patients are known to be alive.”
Image: Graphical illustration of Phase I/II study results. Photo: courtesy of CORCEPT THERAPEUTICS.





