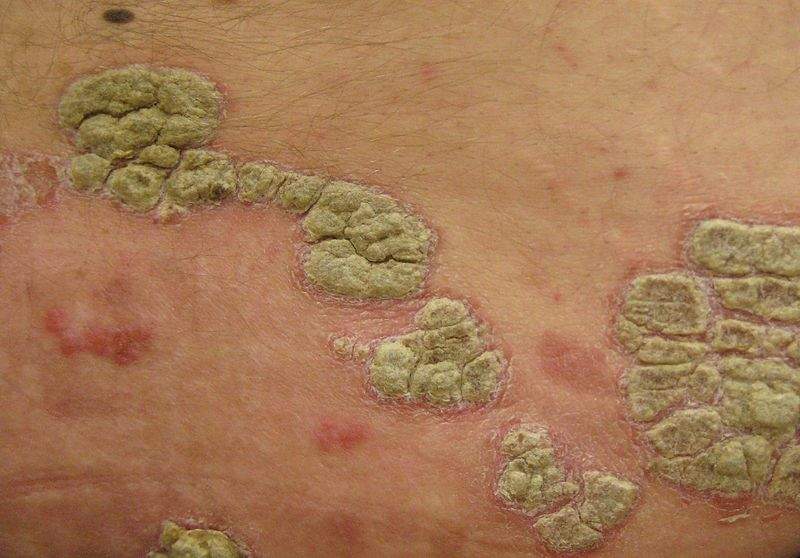
Valeant Pharmaceuticals has reported positive results from a Phase III clinical study to evaluate the safety of IDP-118 (halobetasol propionate and tazarotene) lotion in the treatment of plaque psoriasis.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Within the multi-centre, double-blind, randomised, vehicle-controlled study of 215 adult subjects with moderate-to-severe psoriasis, the lotion showed statistical importance to vehicle with a treatment success rate of 45.33% and a p-value less than 0.001.
The study was carried out for 12 weeks and achieved ‘clear’ to ‘almost clear’ score based on an investigator global assessment (IGA) at eight weeks, and at least two-grade improvement in the IGA at weeks 12, six, four and two as secondary endpoints.
Valeant Pharmaceuticals chairman and CEO Joseph Papa said: "Psoriasis is often difficult to treat, and dealing with this lifelong condition can significantly impact a patient's quality of life.
“Valeant remains committed to continued research into innovative new treatments to improve the lives of those who suffer from psoriasis."
The combination of halobetasol propionate and tazarotene ingredients in IDP-118 with a dual mechanism of action potentially will allow their expanded use with reduced adverse events.
The Phase III programme was preceded by a Phase II study where IDP-118, with a treatment success rate of 52.5%, was believed to be superior to each of the ingredients, as well as the vehicle.
Data from a second confirmatory pivotal Phase III study is expected next year.
Valeant develops, manufactures and markets a range of pharmaceutical products primarily in the areas of dermatology, gastrointestinal disorders, eye health, neurology and branded generics.
Image: Psoriasis. Photo: courtesy of James Heilman, MD.





