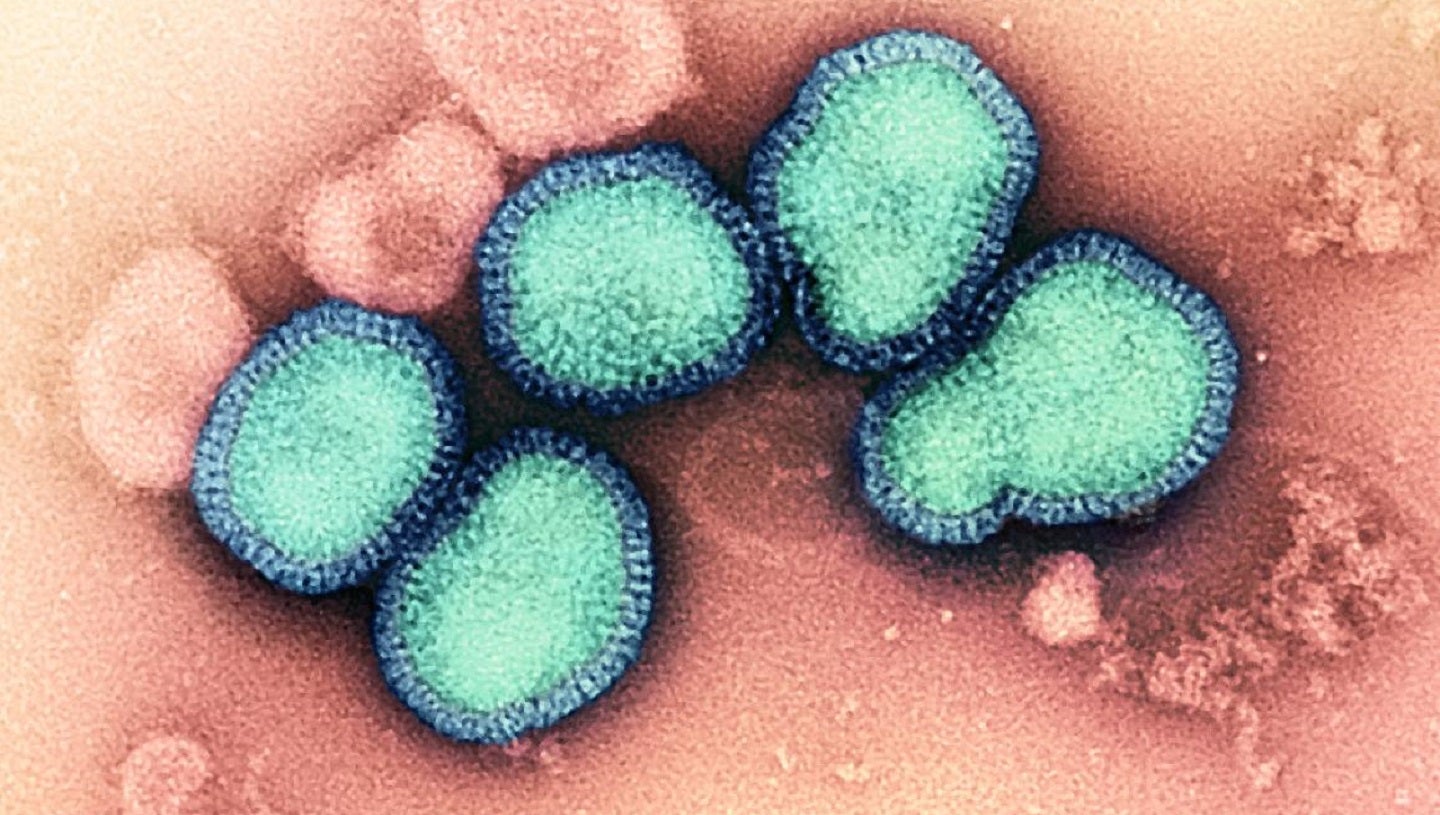
The National Institute of Allergy and Infectious Diseases (NIAID) has started enrolment of volunteers at Duke University to test its experimental vaccine H1ssF-3928 mRNA-LNP against seasonal influenza.
Up to 50 healthy volunteers aged 18 to 49 will be randomised into three groups of ten participants each. Each participant in the three groups will receive 10μg, 25μg and 50μg of the vaccine.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
After evaluation of the optimum dosage, an additional ten subjects will be administered with the same dosage.
The study will compare the immunogenicity and safety of the candidate vaccine against patients who received the current quadrivalent seasonal influenza vaccine.
It will primarily assess the vaccine’s safety and secondarily its efficacy. Participants will be followed for one year after vaccination.
The early-stage trial is being conducted as part of the Collaborative Influenza Vaccine Innovation Centres (CIVICs) programme, created by NIAID in 2019.
NIAID acting director Hugh Auchincloss said: “A universal influenza vaccine would be a major public health achievement and could eliminate the need for both annual development of seasonal influenza vaccines, as well as the need for patients to get a flu shot each year.
“Moreover, some strains of influenza virus have significant pandemic potential. A universal flu vaccine could serve as an important line of defence against the spread of a future flu pandemic.”
The mRNA vaccine uses a specific portion of a flu protein, hemagglutinin (HA), to induce a broad immune response against influenza. HA has also been previously used in a similar vaccine developed by NIAID’s Vaccine Research Center and has shown positive results in early studies.

